Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.4 Relative atomic, molecular and molar mass-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.4 Relative atomic, molecular and molar mass- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.4 Relative atomic, molecular and molar mass- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.4 understand the terms:
i ‘relative atomic mass’ based on the 12C scale
ii ‘relative molecular mass’ and ‘relative formula mass’, including calculating these values from relative atomic masses
The term ‘relative formula mass’ should be used for compounds with giant structures.
iii ‘molar mass’ as the mass per mole of a substance in g mol-1
iv parts per million (ppm), including gases in the atmosphere
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.4 Relative Atomic Mass, Relative Molecular Mass, Relative Formula Mass, Molar Mass, and Parts Per Million
Chemists use relative masses to compare the masses of atoms and molecules. These values are based on a reference scale and are essential for quantitative calculations in chemistry.
Relative Atomic Mass
The relative atomic mass, \( A_r \), of an element is the weighted mean mass of an atom of that element compared with \( \dfrac{1}{12} \) of the mass of a carbon-12 atom.
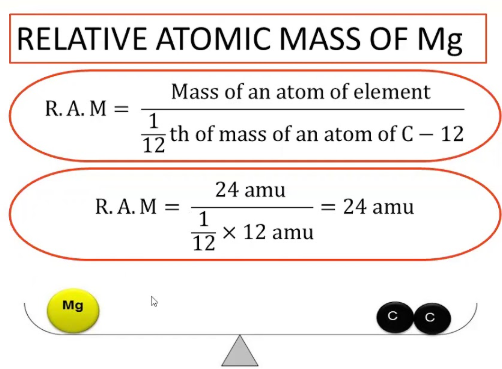
This is known as the carbon-12 scale. The value takes into account the natural abundance of isotopes of the element.
Examples
- \( A_r \) of hydrogen = 1.0
- \( A_r \) of oxygen = 16.0
- \( A_r \) of sodium = 23.0
Relative Molecular Mass
The relative molecular mass, \( M_r \), is the sum of the relative atomic masses of all the atoms in a molecule.
This term is used for substances that exist as molecules.
Example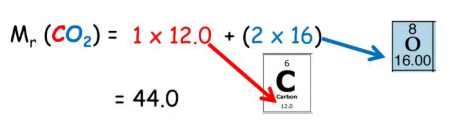
For carbon dioxide \( \mathrm{CO_2} \):
\( M_r = 12 + (2 \times 16) \)
\( M_r = 44 \)
Relative Formula Mass
The relative formula mass is the sum of the relative atomic masses of all atoms in the formula of a compound.
This term is used for compounds that form giant ionic or giant covalent structures, where discrete molecules do not exist.
Example

Molar Mass
The molar mass of a substance is the mass of one mole of that substance, expressed in \( \mathrm{g\ mol^{-1}} \).

The molar mass has the same numerical value as the relative molecular mass or relative formula mass, but includes units.
Examples
- Molar mass of \( \mathrm{H_2O} \) = \( 18\ \mathrm{g\ mol^{-1}} \)
- Molar mass of \( \mathrm{CO_2} \) = \( 44\ \mathrm{g\ mol^{-1}} \)
- Molar mass of \( \mathrm{NaCl} \) = \( 58.5\ \mathrm{g\ mol^{-1}} \)
Parts Per Million (ppm)
Parts per million (ppm) is a unit used to express very small concentrations of substances.
It means the number of parts of a substance present in one million parts of the total mixture.

In atmospheric chemistry, ppm is commonly used to describe the concentration of gases in the air.
Examples
- Carbon dioxide in the atmosphere ≈ 420 ppm
- Trace gases such as methane occur at only a few ppm
Example 1
Calculate the relative molecular mass of sulfuric acid \( \mathrm{H_2SO_4} \).
▶️ Answer/Explanation
Use the relative atomic masses:
\( H = 1,\; S = 32,\; O = 16 \)
\( M_r = (2 \times 1) + 32 + (4 \times 16) \)
\( M_r = 2 + 32 + 64 = 98 \)
Therefore, the relative molecular mass of sulfuric acid is 98.
Example 2
Calculate the molar mass of calcium carbonate \( \mathrm{CaCO_3} \).
▶️ Answer/Explanation
Relative atomic masses:
\( Ca = 40,\; C = 12,\; O = 16 \)
Molar mass \( = 40 + 12 + (3 \times 16) \)
\( = 100\ \mathrm{g\ mol^{-1}} \)
Therefore, the molar mass of calcium carbonate is \( 100\ \mathrm{g\ mol^{-1}} \).
