Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.5 Concentration-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.5 Concentration- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.5 Concentration- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.5 calculate the concentration of a solution in mol dm−3 and g dm−3
Titration calculations are not required at this stage.
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.5 Concentration of Solutions
The concentration of a solution describes how much solute is dissolved in a given volume of solution. In chemistry, concentration is commonly expressed in two ways: mol dm\(^{-3}\) and g dm\(^{-3}\).
At this stage, calculations involving titration are not required. Only basic concentration calculations are needed.
Concentration in \( \mathrm{mol\ dm^{-3}} \)
Concentration in \( \mathrm{mol\ dm^{-3}} \) (moles per cubic decimetre) is the number of moles of solute dissolved in one cubic decimetre of solution.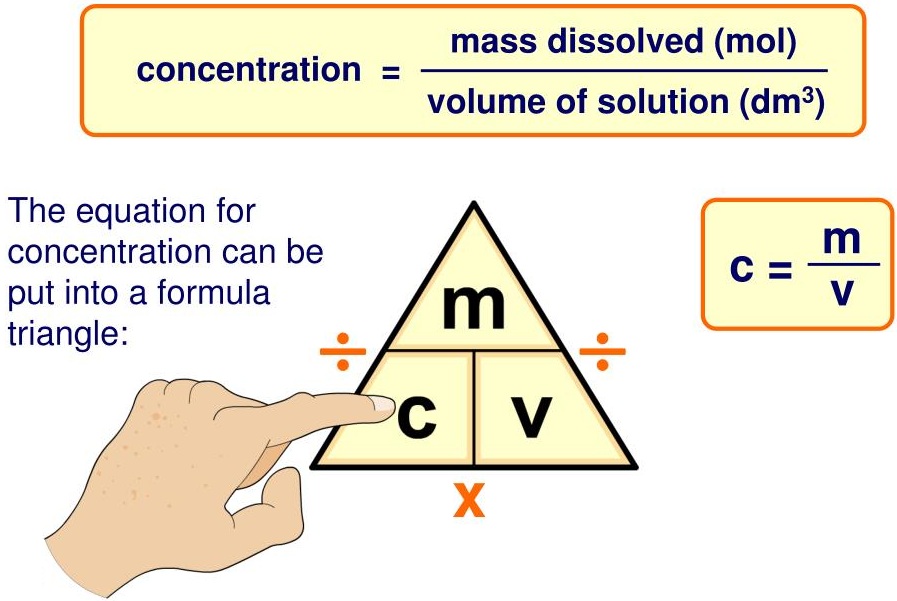
The concentration of a solution can be calculated using the relationship:
\( c = \dfrac{n}{V} \)
where:
- \( c \) = concentration \( (\mathrm{mol\ dm^{-3}}) \)
- \( n \) = number of moles \( (\mathrm{mol}) \)
- \( V \) = volume of solution \( (\mathrm{dm^3}) \)
Example
A solution contains \( 0.50 \) mol of sodium chloride dissolved in \( 2.0\ \mathrm{dm^3} \) of solution. Calculate the concentration.
\( c = \dfrac{0.50}{2.0} \)
\( c = 0.25\ \mathrm{mol\ dm^{-3}} \)
Concentration in \( \mathrm{g\ dm^{-3}} \)
Concentration in \( \mathrm{g\ dm^{-3}} \) is the mass of solute dissolved in one cubic decimetre of solution.
The relationship used is:
\( \text{concentration} = \dfrac{\text{mass of solute}}{\text{volume of solution}} \)
\( c = \dfrac{m}{V} \)
where:
- \( m \) = mass of solute (g)
- \( V \) = volume of solution (dm\(^{3}\))
Example
A solution contains \( 10\ \mathrm{g} \) of sodium chloride dissolved in \( 2.0\ \mathrm{dm^3} \) of solution. Calculate the concentration in \( \mathrm{g\ dm^{-3}} \).
\( c = \dfrac{10}{2.0} \)
\( c = 5.0\ \mathrm{g\ dm^{-3}} \)
Relationship Between the Two Units
Concentration in \( \mathrm{g\ dm^{-3}} \) can be converted to \( \mathrm{mol\ dm^{-3}} \) using the molar mass.
\( c(\mathrm{mol\ dm^{-3}}) = \dfrac{c(\mathrm{g\ dm^{-3}})}{\text{molar mass}} \)
Example 1
Calculate the concentration in \( \mathrm{mol\ dm^{-3}} \) of a solution containing \( 0.20 \) mol of solute in \( 0.50\ \mathrm{dm^3} \).
▶️ Answer/Explanation
\( c = \dfrac{n}{V} \)
\( c = \dfrac{0.20}{0.50} \)
\( c = 0.40\ \mathrm{mol\ dm^{-3}} \)
Example 2
Calculate the concentration in \( \mathrm{g\ dm^{-3}} \) of a solution containing \( 12\ \mathrm{g} \) of solute dissolved in \( 3.0\ \mathrm{dm^3} \) of solution.
▶️ Answer/Explanation
\( c = \dfrac{m}{V} \)
\( c = \dfrac{12}{3.0} \)
\( c = 4.0\ \mathrm{g\ dm^{-3}} \)
