Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.6 Empirical and molecular formulae-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.6 Empirical and molecular formulae- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.6 Empirical and molecular formulae- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.6 be able to use experimental data to calculate empirical and molecular formulae
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.6 Empirical and Molecular Formulae from Experimental Data
Experimental data such as masses or percentages of elements can be used to determine the empirical formula and molecular formula of a compound.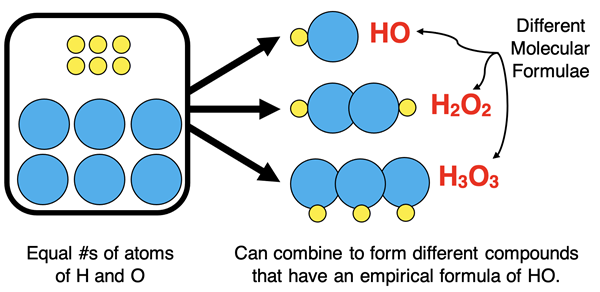
Empirical Formula
The empirical formula shows the simplest whole-number ratio of atoms of each element in a compound.
It is determined using experimental data such as the masses of elements in a compound.
Steps for Calculating an Empirical Formula
- Convert masses of each element to moles.
- Divide each value by the smallest number of moles.
- Simplify the ratio to the smallest whole numbers.
Molecular Formula
The molecular formula shows the actual number of atoms of each element present in a molecule.
It can be determined from the empirical formula if the molar mass of the compound is known.
The relationship between empirical and molecular formulae is:
\( \text{Molecular formula} = (\text{Empirical formula}) \times n \)
\( n = \dfrac{\text{molar mass}}{\text{empirical formula mass}} \)
Example 1
A compound contains \( 2.4\ \mathrm{g} \) of carbon and \( 0.4\ \mathrm{g} \) of hydrogen. Determine the empirical formula of the compound.
▶️ Answer/Explanation
Step 1: Convert masses to moles
\( n(C) = \dfrac{2.4}{12} = 0.20 \)
\( n(H) = \dfrac{0.4}{1} = 0.40 \)
Step 2: Divide by the smallest value
C : H = \( 0.20 : 0.40 \)
C : H = \( 1 : 2 \)
Therefore the empirical formula is
\( \mathrm{CH_2} \)
Example 2
A compound has an empirical formula \( \mathrm{CH_2O} \) and a molar mass of \( 180\ \mathrm{g\ mol^{-1}} \). Determine the molecular formula.
▶️ Answer/Explanation
Step 1: Calculate empirical formula mass
\( M_r = 12 + 2(1) + 16 = 30 \)
Step 2: Calculate multiplier
\( n = \dfrac{180}{30} = 6 \)
Step 3: Multiply empirical formula
\( (\mathrm{CH_2O}) \times 6 \)
\( \mathrm{C_6H_{12}O_6} \)
Therefore the molecular formula is \( \mathrm{C_6H_{12}O_6} \).
