Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.7 Reacting masses calculations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.7 Reacting masses calculations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.7 Reacting masses calculations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.7 be able to use chemical equations to calculate reacting masses and vice versa, using the concepts of amount of substance and molar mass
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.7 Reacting Mass Calculations
Balanced chemical equations show the ratio in which substances react. Using the concepts of amount of substance (moles) and molar mass, it is possible to calculate the mass of one substance that reacts with or produces another substance.
These calculations are called reacting mass calculations or stoichiometric calculations.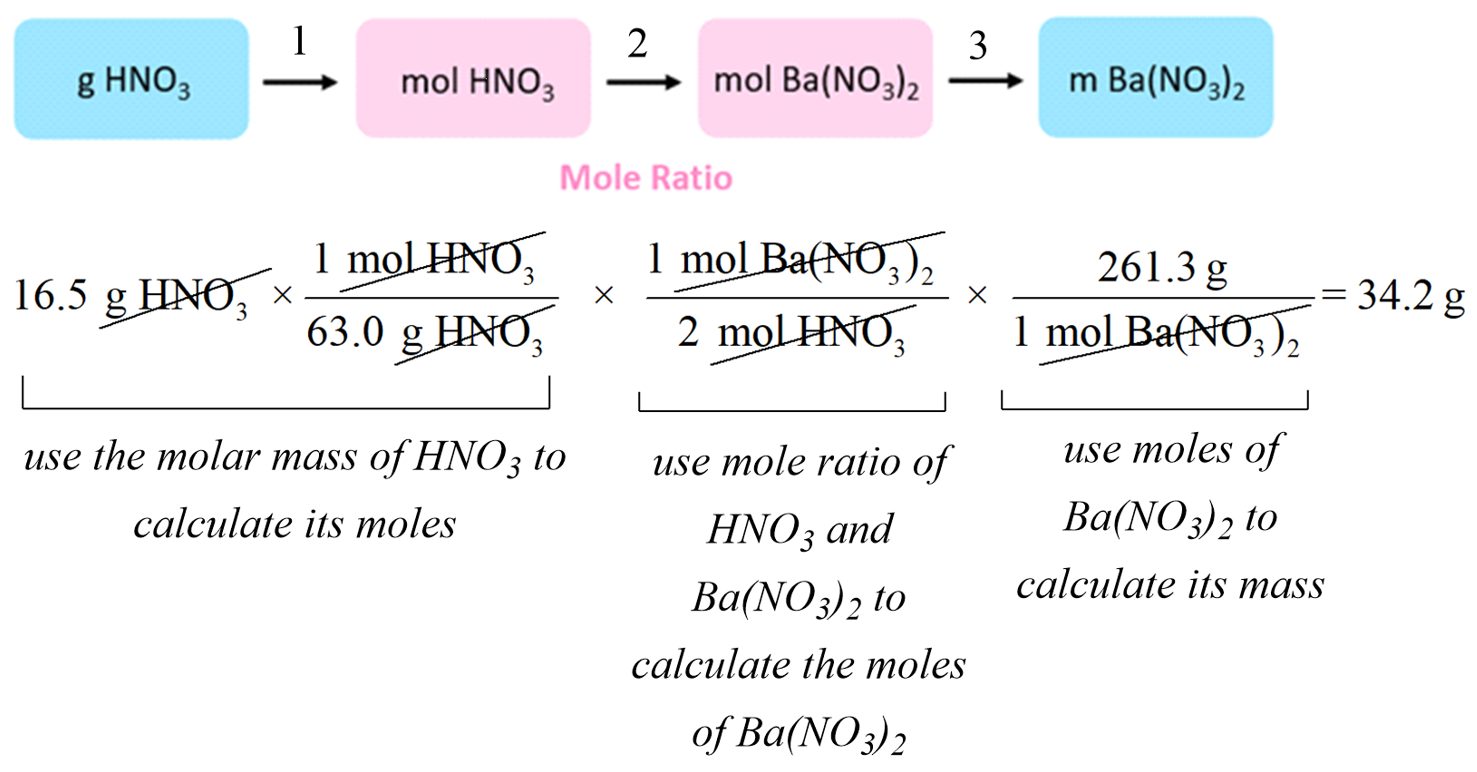
Key Relationships
\( n = \dfrac{m}{M} \)
where:
- \( n \) = number of moles
- \( m \) = mass (g)
- \( M \) = molar mass \( (\mathrm{g\ mol^{-1}}) \)
The balanced chemical equation provides the mole ratio between reactants and products.
General Steps for Reacting Mass Calculations
- Write the balanced chemical equation.
- Convert the known mass into moles.
- Use the mole ratio from the equation to find moles of the required substance.
- Convert moles back to mass if required.
Example 1
Calculate the mass of magnesium oxide produced when \( 4.8\ \mathrm{g} \) of magnesium reacts with excess oxygen.
Balanced equation:
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
▶️ Answer/Explanation
Step 1: Calculate moles of magnesium
\( n = \dfrac{m}{M} \)
\( n = \dfrac{4.8}{24} = 0.20\ \mathrm{mol} \)
Step 2: Use mole ratio
From the equation:
\( Mg : MgO = 1 : 1 \)
Moles of \( MgO = 0.20\ \mathrm{mol} \)
Step 3: Convert to mass
Molar mass of \( MgO = 24 + 16 = 40 \)
\( m = nM \)
\( m = 0.20 \times 40 = 8.0\ \mathrm{g} \)
Therefore \( 8.0\ \mathrm{g} \) of magnesium oxide is produced.
Example 2
What mass of carbon dioxide is produced when \( 12\ \mathrm{g} \) of carbon reacts completely with oxygen?
Balanced equation:
\( \mathrm{C + O_2 \rightarrow CO_2} \)
▶️ Answer/Explanation
Step 1: Calculate moles of carbon
\( n = \dfrac{12}{12} = 1.0\ \mathrm{mol} \)
Step 2: Use mole ratio
\( C : CO_2 = 1 : 1 \)
Moles of \( CO_2 = 1.0\ \mathrm{mol} \)
Step 3: Convert to mass
Molar mass of \( CO_2 = 12 + 32 = 44 \)
\( m = 1.0 \times 44 = 44\ \mathrm{g} \)
Therefore \( 44\ \mathrm{g} \) of carbon dioxide is produced.
