Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.9 Percentage yield and atom economy-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.9 Percentage yield and atom economy- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.9 Percentage yield and atom economy- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
be able to calculate percentage yields and percentage atom economies (by mass) in laboratory and industrial processes, using chemical equations and experimental results![]()
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.9 Percentage Yield and Atom Economy
In chemical reactions, the amount of product obtained in practice is often different from the theoretical amount predicted by the balanced chemical equation. Chemists therefore use percentage yield and atom economy to measure the efficiency of reactions in laboratory and industrial processes.
Percentage Yield
Percentage yield compares the actual amount of product obtained with the theoretical amount predicted by calculations.

The theoretical yield is the maximum possible amount of product calculated from the balanced equation. The actual yield is the amount actually obtained from the experiment.
Percentage yield is usually less than 100% because:
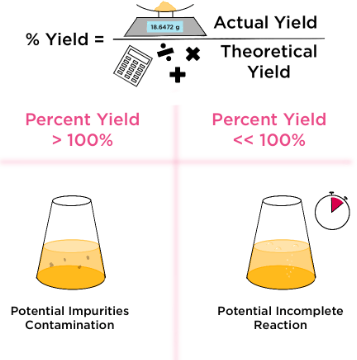
- some product may be lost during separation or purification
- the reaction may not go to completion
- side reactions may produce unwanted products
Atom Economy
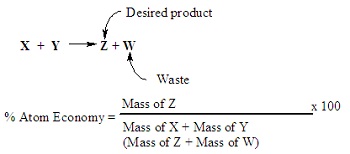
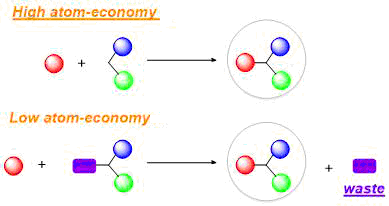
Atom economy measures how efficiently atoms in the reactants are converted into the desired product.
\( \text{Atom economy} = \dfrac{\text{molar mass of desired product}}{\text{sum of molar masses of all products}} \times 100\% \)
A reaction with a high atom economy produces fewer unwanted by-products and is therefore more efficient and environmentally friendly.
Example 1
In a reaction, the theoretical yield of a product is \( 12.0\ \mathrm{g} \), but the experiment produces \( 9.6\ \mathrm{g} \). Calculate the percentage yield.
▶️ Answer/Explanation
\( \text{Percentage yield} = \dfrac{9.6}{12.0} \times 100 \)
\( = 80\% \)
Therefore, the percentage yield of the reaction is 80%.
Example 2
Calculate the atom economy for the reaction:
\( \mathrm{C_2H_4 + H_2O \rightarrow C_2H_5OH} \)
Assume ethanol is the desired product.
▶️ Answer/Explanation
Step 1: Calculate molar mass of ethanol
\( M_r(\mathrm{C_2H_5OH}) = 46 \)
Step 2: Calculate total molar mass of products
Total \( M_r = 46 \)
Step 3: Calculate atom economy
\( \text{Atom economy} = \dfrac{46}{46} \times 100 \)
\( = 100\% \)
Therefore, the atom economy of this reaction is 100%.
