Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.1–2.2 Structure of the atom; relative mass and charge-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.1–2.2 Structure of the atom; relative mass and charge- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.1–2.2 Structure of the atom; relative mass and charge- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.1 know the structure of an atom in terms of electrons, protons and neutrons
2.2 know the relative mass and charge of protons, neutrons and electrons
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.1 Structure of the Atom
All atoms are made up of three fundamental subatomic particles: protons, neutrons, and electrons. The arrangement of these particles determines the structure of the atom.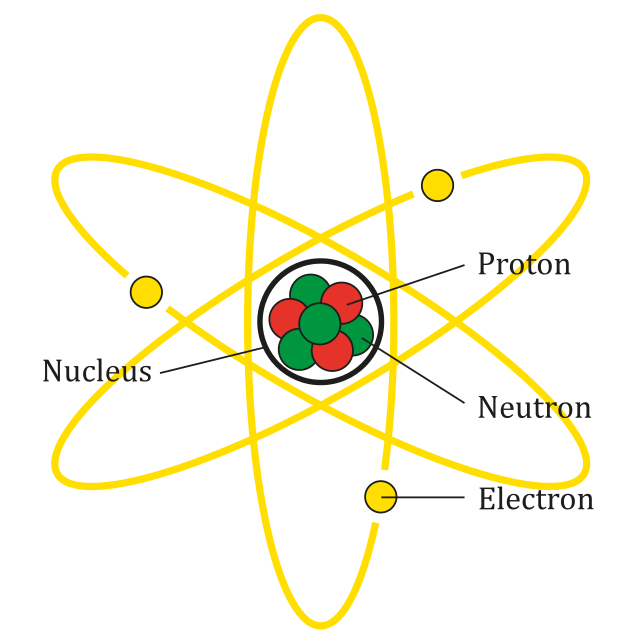
Nucleus
The nucleus is the small, dense central region of an atom that contains protons and neutrons.
Almost all the mass of the atom is concentrated in the nucleus because protons and neutrons are much heavier than electrons.
Protons
Protons are positively charged particles found in the nucleus of the atom.
The number of protons in an atom determines the identity of the element. This number is called the atomic number.
Neutrons
Neutrons are neutral particles (no charge) that are also found in the nucleus of the atom.
Neutrons contribute to the mass of the atom and help stabilise the nucleus.
Electrons
Electrons are negatively charged particles that move in shells or energy levels around the nucleus.
Electrons occupy regions of space called orbitals and are responsible for chemical bonding and reactions.
Overall Structure
An atom therefore consists of:
- A dense nucleus containing protons and neutrons
- Electrons arranged in shells surrounding the nucleus
In a neutral atom, the number of electrons is equal to the number of protons.
Example :
A carbon atom contains 6 protons, 6 neutrons, and 6 electrons. Identify where each of these particles is located in the atom.
▶️ Answer/Explanation
Protons → located in the nucleus.
Neutrons → located in the nucleus.
Electrons → move in shells surrounding the nucleus.
Conclusion: The nucleus contains protons and neutrons, while electrons occupy energy levels around the nucleus.
Example :
An atom has 11 protons, 12 neutrons, and 11 electrons. Describe the structure of this atom.
▶️ Answer/Explanation
The nucleus contains 11 protons and 12 neutrons.
The 11 electrons move in shells around the nucleus.
Since the number of protons equals the number of electrons, the atom is electrically neutral.
2.2 Relative Mass and Charge of Subatomic Particles
Atoms contain three main subatomic particles: protons, neutrons, and electrons. Each particle has a characteristic relative mass and electrical charge.
These properties determine the structure of atoms and explain why most of the mass of an atom is concentrated in the nucleus.

Relative Mass and Charge
| Particle | Relative Mass | Relative Charge | Location |
|---|---|---|---|
| Proton | 1 | +1 | Nucleus |
| Neutron | 1 | 0 | Nucleus |
| Electron | 1/1836 | −1 | Electron shells |
Key Points
- Protons and neutrons have almost identical relative masses.
- Electrons have a much smaller mass compared with protons and neutrons.
- Most of the mass of the atom is concentrated in the nucleus.
- In a neutral atom, the number of protons equals the number of electrons.
Example :
An atom contains 15 protons, 16 neutrons and 15 electrons.
(a) Determine the total relative mass of the atom using the relative masses of subatomic particles.
(b) Explain why the electrons contribute very little to the mass of the atom.
▶️ Answer/Explanation
(a) Total relative mass
Relative mass of proton = 1 Relative mass of neutron = 1
Mass from protons = 15 × 1 = 15
Mass from neutrons = 16 × 1 = 16
Total ≈ 31
(b) Electron contribution
The relative mass of an electron is only \( \frac{1}{1836} \). Therefore the mass of electrons is extremely small compared with protons and neutrons.
As a result, almost all the mass of the atom is concentrated in the nucleus.
Example :
An ion of magnesium has the following particles:
12 protons, 12 neutrons, 10 electrons
(a) Determine the overall charge of the ion.
(b) Explain how this charge arises from the relative charges of the particles.
▶️ Answer/Explanation
(a) Overall charge
Charge from protons = \( +12 \)
Charge from electrons = \( -10 \)
Total charge = \( +12 – 10 = +2 \)
Therefore the ion is \( \mathrm{Mg^{2+}} \).
(b) Explanation
The ion has more protons than electrons. Because protons carry a +1 charge and electrons carry a −1 charge, the imbalance results in an overall positive charge.
