Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.10 Factors affecting ionisation energy-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.10 Factors affecting ionisation energy- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.10 Factors affecting ionisation energy- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.10 understand how ionisation energies are influenced by the number of protons in the nucleus, the electron shielding and the sub-shell from which the electron is removed
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.10 Factors Affecting Ionisation Energy
Ionisation energy depends on the strength of attraction between the positively charged nucleus and the negatively charged outermost electron. The stronger this attraction, the more energy is required to remove the electron.
Three key factors influence this attraction: nuclear charge (number of protons), electron shielding, and the sub-shell (orbital type) from which the electron is removed.
1. Nuclear Charge (Number of Protons)
The nuclear charge refers to the total positive charge of the nucleus, which depends on the number of protons present.
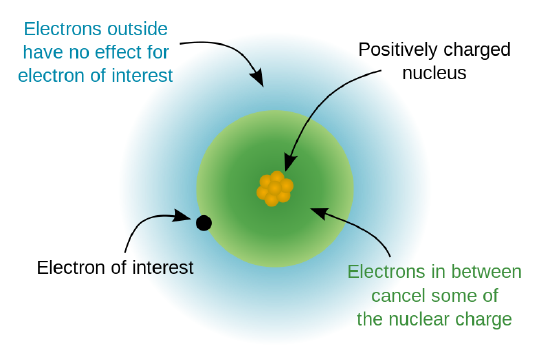
As the number of protons increases, the positive charge of the nucleus increases. This leads to a stronger electrostatic attraction between the nucleus and the outer electrons.
A stronger attraction means that more energy is required to remove an electron, so the ionisation energy increases.
For example, across a period in the periodic table, the number of protons increases while electrons are added to the same shell. This results in a gradual increase in ionisation energy.
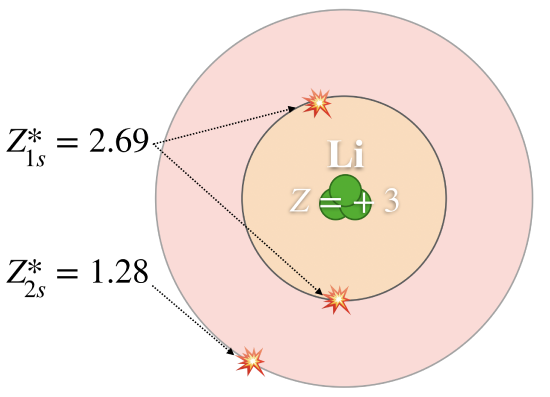
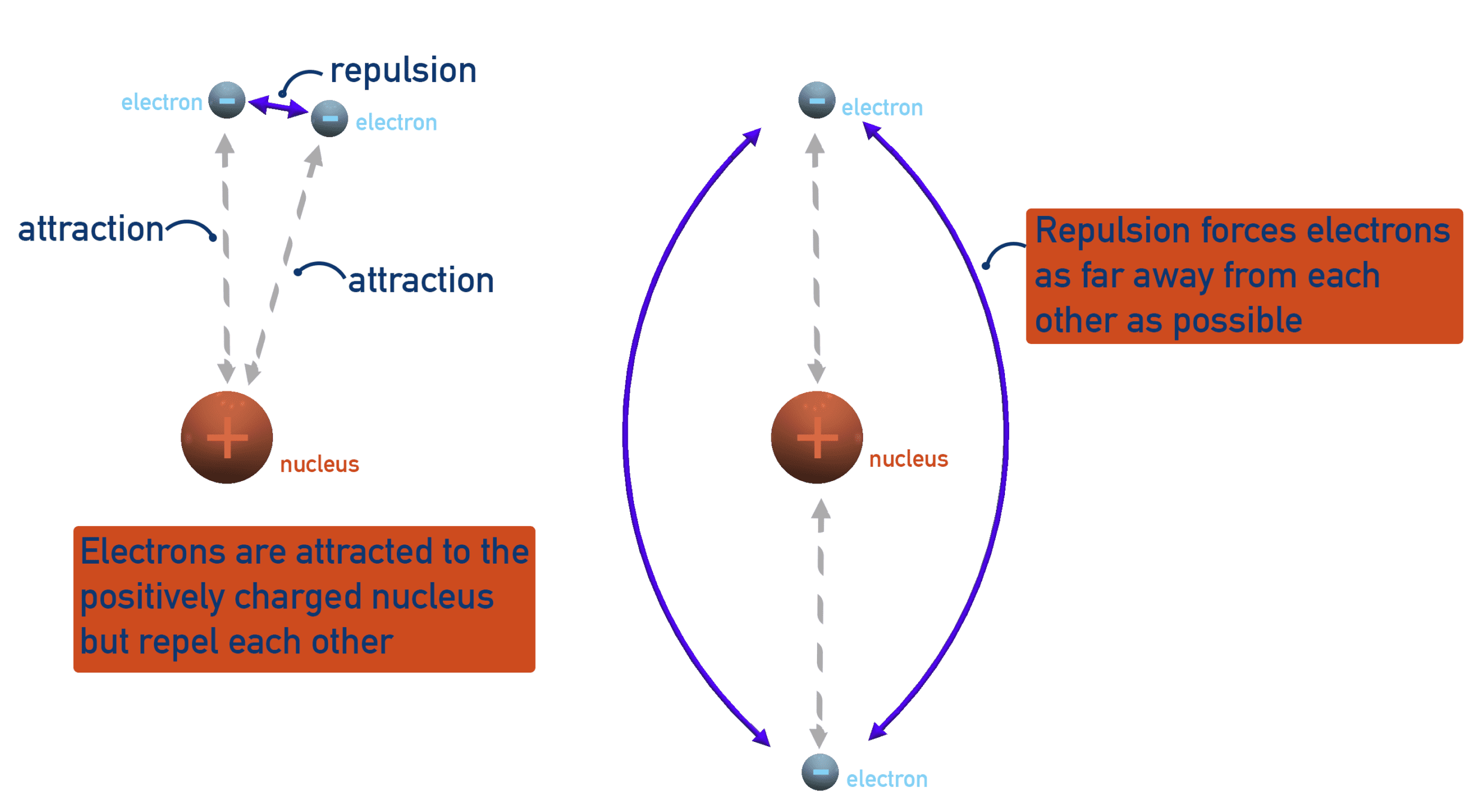
2. Electron Shielding
Electron shielding occurs when inner-shell electrons repel outer-shell electrons, reducing the effective attraction between the nucleus and the outermost electron.
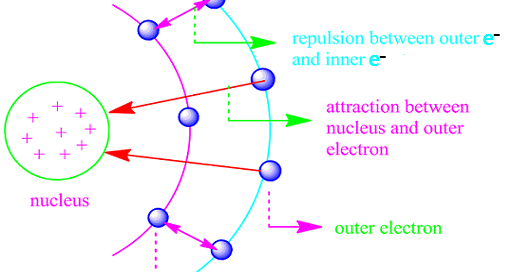
Inner electrons act as a barrier between the nucleus and the outer electrons. As a result, the outer electrons do not experience the full nuclear charge.
This reduced attraction is known as the effective nuclear charge.
As shielding increases (for example, down a group), the outer electrons are less strongly attracted to the nucleus, so less energy is required to remove them. Therefore, ionisation energy decreases.
This explains why elements lower down a group have lower ionisation energies than those above them.
3. Distance from the Nucleus
Electrons that are further from the nucleus experience weaker electrostatic attraction due to increased distance and shielding.
As atomic size increases, the outer electrons are located further away from the nucleus, making them easier to remove.
Therefore, greater distance from the nucleus leads to lower ionisation energy.
4. Sub-shell (Orbital Type)
Electrons occupy different types of orbitals (sub-shells), such as s and p, which have slightly different energy levels.
Electrons in s orbitals are closer to the nucleus and experience stronger attraction. Electrons in p orbitals are slightly further away and more shielded.
As a result, electrons in p orbitals require less energy to remove compared to electrons in s orbitals of the same shell.
This explains anomalies in ionisation energy trends, such as:
Magnesium (3s²) has a higher ionisation energy than aluminium (3p¹)
Summary
- Higher nuclear charge → stronger attraction → higher ionisation energy
- More shielding → weaker attraction → lower ionisation energy
- Greater distance → weaker attraction → lower ionisation energy
- p electrons are easier to remove than s electrons
Example 1:
Explain the trend in first ionisation energy across Period 3 from sodium to argon.
▶️ Answer/Explanation
Across Period 3, the number of protons increases, leading to a higher nuclear charge.
Electrons are added to the same shell, so shielding remains relatively constant.
Therefore, the attraction between the nucleus and the outer electron increases, causing ionisation energy to increase across the period.
However, there are small decreases between magnesium and aluminium (due to p orbital) and between phosphorus and sulfur (due to electron pairing repulsion).
Example 2:
Explain why the first ionisation energy of potassium is lower than that of sodium.
▶️ Answer/Explanation
Potassium has more electron shells than sodium, so its outer electron is further from the nucleus.
There is also greater electron shielding due to additional inner shells.
Although potassium has more protons, the increased distance and shielding reduce the attraction between the nucleus and the outer electron.
Therefore, less energy is required to remove the electron, resulting in a lower ionisation energy.
