Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.11 Successive IE and sub-shell evidence-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.11 Successive IE and sub-shell evidence- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.11 Successive IE and sub-shell evidence- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.11 know that ideas about electronic structure developed from:
i an understanding that successive ionisation energies provide evidence for the existence of quantum shells and the group to which the element belongs
ii an understanding that the first ionisation energy of successive elements provides evidence for electron sub-shells
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.11 Development of Electronic Structure from Ionisation Energies
The modern model of the atom, including the arrangement of electrons into shells and sub-shells, was developed through careful analysis of ionisation energy data.
By studying both successive ionisation energies and first ionisation energies across a period, scientists were able to deduce how electrons are organised within atoms.
(i) Evidence for Quantum Shells from Successive Ionisation Energies
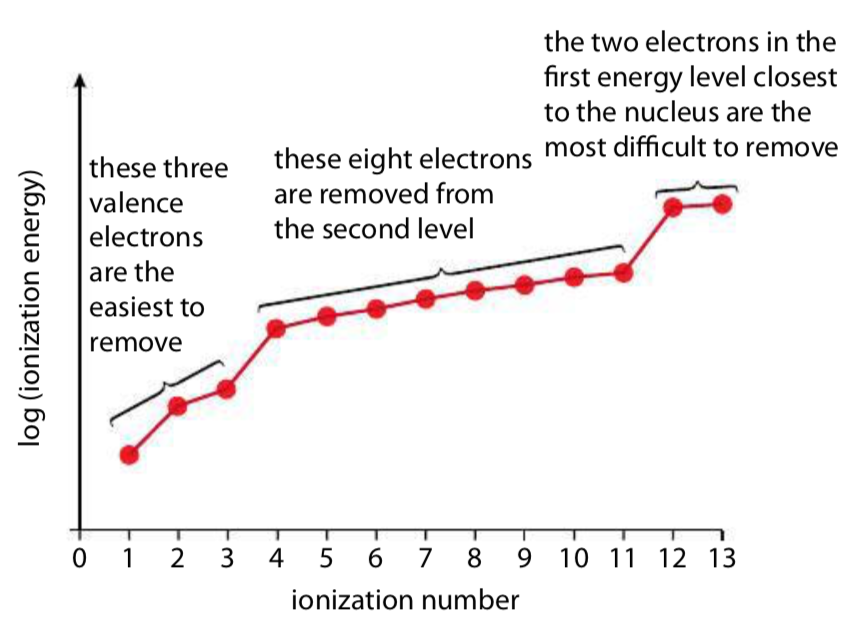
Successive ionisation energies refer to the energy required to remove electrons one at a time from the same atom in the gaseous state.
When these values are analysed, a characteristic pattern is always observed:
A steady increase in ionisation energy followed by a very large jump.
The steady increase occurs because after each electron is removed, the ion becomes more positively charged. This increases the attraction between the nucleus and the remaining electrons, so more energy is required for each successive removal.
The large jump is the key piece of evidence for quantum shells.
This jump occurs when all electrons in the outer shell have been removed and the next electron is removed from an inner shell.
Inner-shell electrons are much closer to the nucleus and experience significantly less shielding, so they are much more strongly attracted to the nucleus.
As a result, a much larger amount of energy is required to remove an inner-shell electron.
This provides strong evidence that electrons are arranged in distinct energy levels (shells) rather than being evenly distributed.
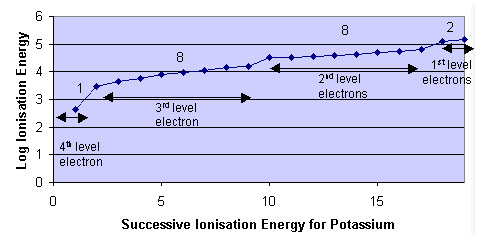
Furthermore, the number of electrons removed before the large jump corresponds to the number of electrons in the outer shell.
This allows the group of the element to be determined, since elements in the same group have the same number of outer-shell electrons.
(ii) Evidence for Sub-shells from First Ionisation Energies Across a Period
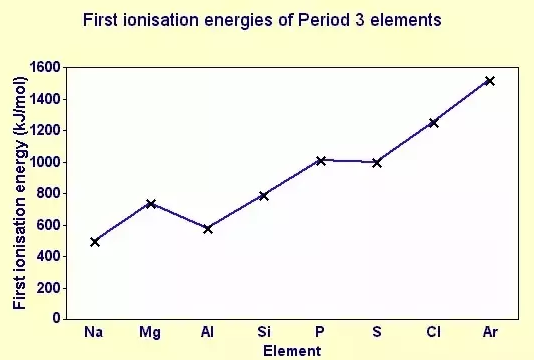
When the first ionisation energies of elements across a period are plotted, a general increase is observed.
This increase is due to:
Increasing nuclear charge (more protons), while electrons are added to the same shell, so shielding remains similar.
However, the graph is not perfectly smooth. There are small but important decreases in ionisation energy at certain points.
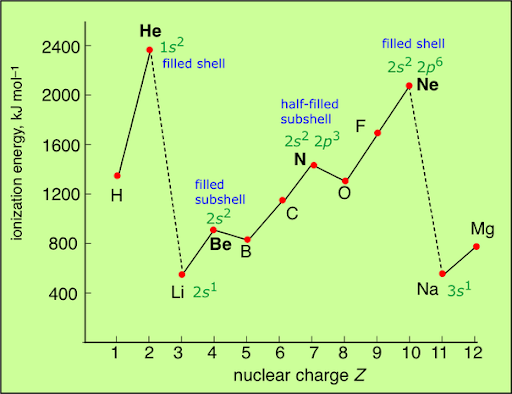
These irregularities provide evidence for the existence of sub-shells.
The first key irregularity occurs when electrons begin to occupy a p sub-shell after filling an s sub-shell.
- Electrons in p orbitals are higher in energy and further from the nucleus than electrons in s orbitals of the same shell.
- They are also slightly more shielded, so they experience weaker attraction and are easier to remove.
The second irregularity occurs when electrons begin to pair within the same orbital.
- When two electrons occupy the same orbital, they repel each other due to their negative charges.
- This electron-electron repulsion reduces the energy required to remove one of the electrons.
These observations show that:
Each shell is divided into sub-levels (sub-shells) with slightly different energies, and that electron arrangement within orbitals affects stability.
Example 1:
The successive ionisation energies of an element are:
IE₁ = 590 IE₂ = 1820 IE₃ = 2750 IE₄ = 11600 (all values in kJ mol\(^{-1}\))
(a) Identify the position of the large jump. (b) Explain what this indicates about electron shells. (c) Deduce the group of the element.
▶️ Answer/Explanation
(a)
The large jump occurs between IE₃ and IE₄.
(b)
This indicates that the first three electrons are removed from the same outer shell, while the fourth electron is removed from an inner shell.
This provides evidence for the existence of discrete electron shells.
(c)
Since three electrons are removed before the large jump, the atom has 3 outer-shell electrons.
Therefore, the element belongs to Group 3 (Group 13).
Example 2:
Across Period 3, the first ionisation energy generally increases, but decreases between magnesium → aluminium and phosphorus → sulfur.
Explain how these decreases provide evidence for sub-shells and electron arrangement.
▶️ Answer/Explanation
The decrease from magnesium to aluminium occurs because the electron removed from aluminium is in a 3p orbital, whereas in magnesium it is in a 3s orbital.
The 3p orbital is higher in energy, further from the nucleus, and more shielded, so the electron is easier to remove.
This provides evidence that sub-shells exist within the same shell and have different energies.
The decrease from phosphorus to sulfur occurs because sulfur has a pair of electrons in one p orbital.
The repulsion between paired electrons makes it easier to remove one of them.
This shows that electron pairing within orbitals affects energy and stability, providing further evidence for sub-shell structure.
