Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.13 Orbital filling and spin-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.13 Orbital filling and spin- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.13 Orbital filling and spin- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.13 know that orbitals in sub-shells:
i each take a single electron before pairing up
ii pair up with two electrons of opposite spin
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.13 Electron Arrangement in Orbitals
Electrons are arranged within orbitals according to a set of fundamental rules that determine how they occupy available energy levels and sub-shells. These rules explain how electrons fill orbitals and why certain arrangements are more stable than others.
Two key principles describe how electrons occupy orbitals within a sub-shell:
(i) orbitals are singly occupied before any pairing occurs
(ii) electrons in the same orbital must have opposite spins
(i) Single Occupancy of Orbitals (Hund’s Rule)
Electrons occupy available orbitals singly before any pairing takes place.
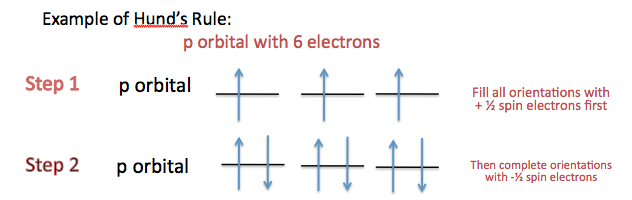
This is known as Hund’s Rule. It applies particularly to sub-shells that contain more than one orbital, such as the p sub-shell (which contains three orbitals).
When electrons are placed into orbitals of equal energy (degenerate orbitals), they spread out as much as possible, occupying separate orbitals first.
This arrangement minimises electron-electron repulsion because electrons are kept as far apart as possible.
As a result, the atom is in a lower energy and more stable state.
For example, in a p sub-shell with three orbitals:
Correct arrangement for three electrons: \( \uparrow \; \uparrow \; \uparrow \)
Electrons only begin to pair once all orbitals contain one electron.
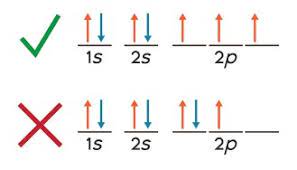
(ii) Pairing of Electrons with Opposite Spin (Pauli Exclusion Principle)
An orbital can hold a maximum of two electrons, and these electrons must have opposite spins.
This is described by the Pauli Exclusion Principle, which states that no two electrons in an atom can have the same set of quantum numbers.
When two electrons occupy the same orbital, they are paired and must spin in opposite directions to reduce repulsion and maintain stability.
This is commonly represented using arrows:
\( \uparrow \downarrow \)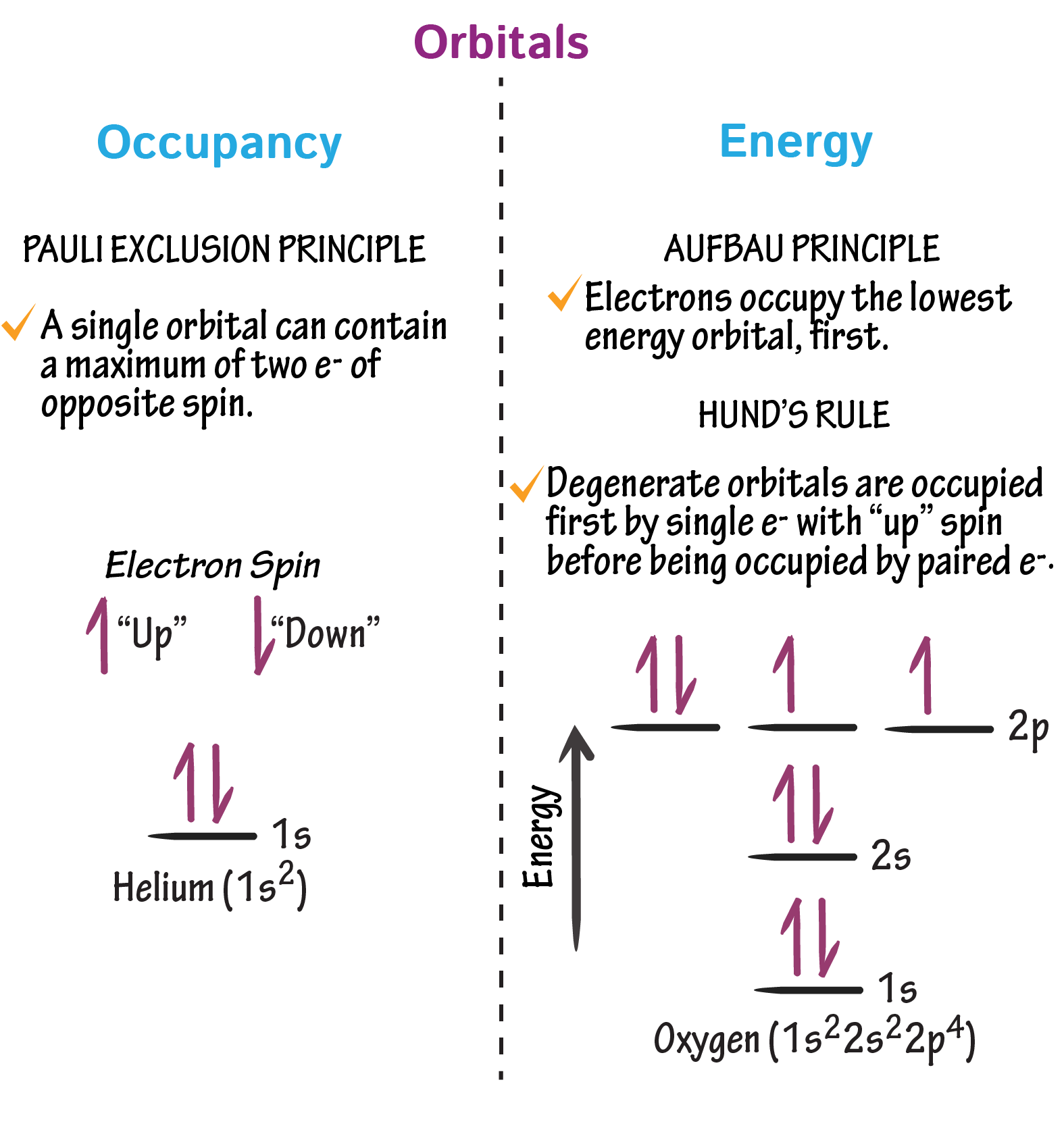
If electrons in the same orbital had the same spin, they would repel each other more strongly and violate the Pauli Exclusion Principle.
Combined Effect of Both Rules
Together, Hund’s Rule and the Pauli Exclusion Principle determine the correct electron arrangement within sub-shells.
For example, in a p sub-shell:
- 3 electrons → \( \uparrow \; \uparrow \; \uparrow \)
- 4 electrons → \( \uparrow\downarrow \; \uparrow \; \uparrow \)
- 6 electrons → \( \uparrow\downarrow \; \uparrow\downarrow \; \uparrow\downarrow \)
This systematic filling pattern is essential for understanding electron configurations and chemical behaviour.
Example 1:
Draw the arrangement of electrons in the 2p sub-shell for nitrogen (\( 2p^3 \)) and oxygen (\( 2p^4 \)).
▶️ Answer/Explanation
Nitrogen (2p³)
\( \uparrow \; \uparrow \; \uparrow \)
Each orbital contains one electron, following Hund’s Rule.
Oxygen (2p⁴)
\( \uparrow\downarrow \; \uparrow \; \uparrow \)
After each orbital is singly occupied, the fourth electron pairs in one orbital.
This introduces electron repulsion, which helps explain trends in ionisation energy.
Example 2:
Explain why paired electrons in the same orbital are easier to remove than unpaired electrons in separate orbitals.
▶️ Answer/Explanation
When two electrons occupy the same orbital, they are in close proximity and repel each other due to their negative charges.
This electron-electron repulsion increases the energy of the electrons, making them less stable.
As a result, less energy is required to remove one of the paired electrons compared to an unpaired electron in a separate orbital.
This explains observations such as the lower ionisation energy of sulfur compared to phosphorus.
