Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.14–2.15 Electron configuration and chemical properties-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.14–2.15 Electron configuration and chemical properties- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.14–2.15 Electron configuration and chemical properties- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.14 be able to predict the electronic configuration of atoms of the elements from hydrogen to krypton inclusive and their ions, using s, p, d notation and electron-inboxes notation
2.15 understand that electronic configuration determines the chemical properties of an element
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.14 Electronic Configurations (s, p, d notation and Electron-in-Boxes)
The electronic configuration of an atom describes how electrons are arranged in orbitals. This arrangement follows a set of rules based on energy levels, sub-shells, and orbital filling order.
You must be able to write electronic configurations for elements from hydrogen to krypton, and also for their ions, using both:
• s, p, d notation • electron-in-boxes (orbital box) notation
Order of Filling (Aufbau Principle)
Electrons fill orbitals starting from the lowest energy level first.
The order of filling is:
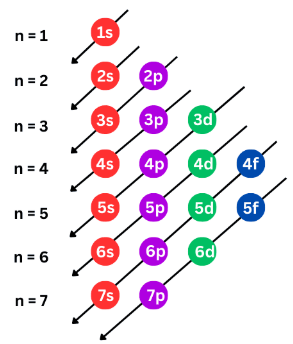
Each sub-shell has a maximum number of electrons:
s = 2 electrons p = 6 electrons d = 10 electrons
Rules for Filling Orbitals
- Electrons fill lowest energy orbitals first (Aufbau Principle)
- Orbitals are singly occupied before pairing (Hund’s Rule)
- Each orbital holds a maximum of two electrons with opposite spin (Pauli Exclusion Principle)
s, p, d Notation
Electronic configuration is written by listing sub-shells and the number of electrons in each:
Example (oxygen, 8 electrons): \( 1s^2 \; 2s^2 \; 2p^4 \)
Example (calcium, 20 electrons):
\( 1s^2 \; 2s^2 \; 2p^6 \; 3s^2 \; 3p^6 \; 4s^2 \)
Electron-in-Boxes (Orbital) Notation
Orbitals can be represented as boxes, with arrows showing electron spin.
Example (oxygen, \( 2p^4 \)):
[↑↓] [↑↓] [↑ ] [↑ ]
Electrons occupy separate orbitals before pairing, following Hund’s Rule.
Electronic Configurations of Ions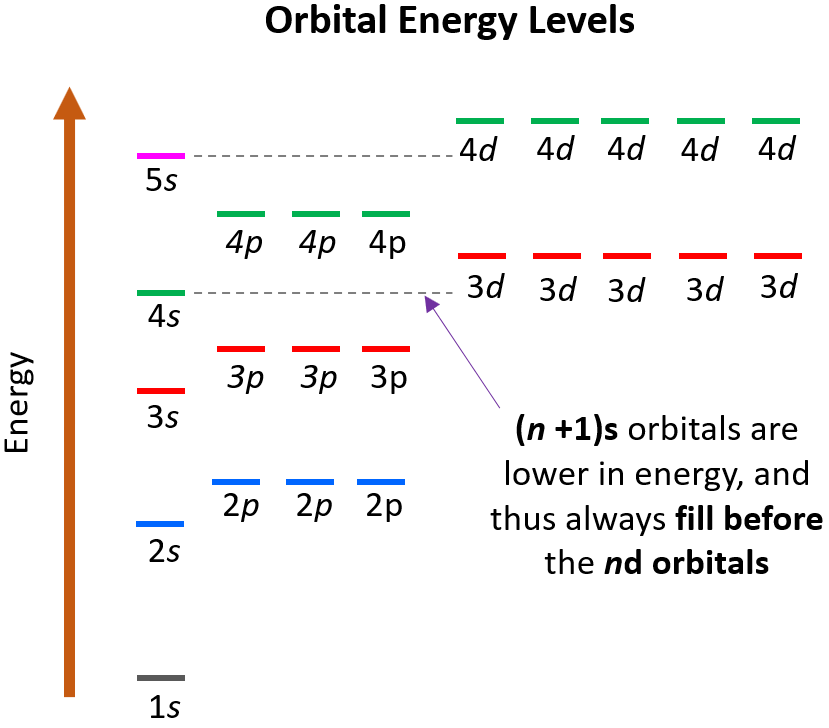
When atoms form ions, electrons are removed or added.
Key rule:
Electrons are removed from the highest energy level (outermost shell) first.
Example (sodium):
- Na: \( 1s^2 2s^2 2p^6 3s^1 \)
- Na\( ^+ \): \( 1s^2 2s^2 2p^6 \)
Example (iron):
- Fe: \( [Ar] 4s^2 3d^6 \)
- Fe\( ^{2+} \): \( [Ar] 3d^6 \)
Note that electrons are removed from 4s before 3d.
Example 1:
Write the electronic configuration of phosphorus (Z = 15) in:
(a) s, p notation (b) electron-in-boxes notation for the 3p sub-shell
▶️ Answer/Explanation
(a)
\( 1s^2 2s^2 2p^6 3s^2 3p^3 \)
(b)
3p: [↑] [↑] [↑]
Electrons occupy separate orbitals before pairing (Hund’s Rule).
Example 2:
Write the electronic configuration of the ion \( \mathrm{Fe^{3+}} \).
▶️ Answer/Explanation
Fe: \( [Ar] 4s^2 3d^6 \)
Remove electrons from the outermost shell first (4s), then from 3d:
Fe\(^{3+}\): \( [Ar] 3d^5 \)
This results in a half-filled d sub-shell, which is relatively stable.
2.15 Electronic Configuration and Chemical Properties
The electronic configuration of an element determines how its electrons are arranged in shells and sub-shells. This arrangement directly influences how an atom behaves chemically, including how it forms bonds, reacts with other substances, and the types of ions it forms.
In particular, the electrons in the outer shell (valence electrons) play the most important role in determining chemical properties.
Role of Outer-Shell Electrons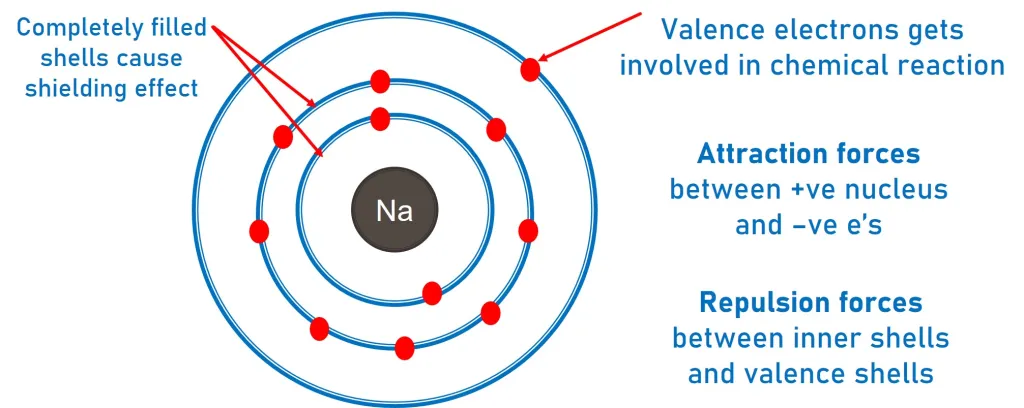
Chemical reactions involve the transfer or sharing of electrons, and these electrons are almost always from the outer shell.
The number of outer-shell electrons determines:
- the reactivity of the element
- the type of bonding it forms (ionic or covalent)
- the charge of ions it is likely to form
Elements tend to react in ways that allow them to achieve a more stable electronic configuration, often similar to that of a noble gas.
Elements in the Same Group
Elements in the same group of the periodic table have the same number of outer-shell electrons.
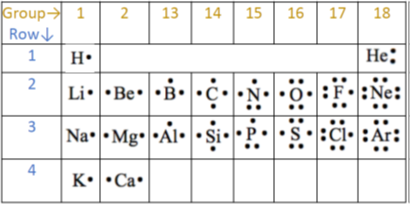
Because of this, they have similar chemical properties and react in similar ways.
For example:
- Group 1 elements all have one outer electron and tend to lose this electron to form \( +1 \) ions.
- Group 7 elements have seven outer electrons and tend to gain one electron to form \( -1 \) ions.
Formation of Ions
Atoms form ions by losing or gaining electrons to achieve a more stable configuration.
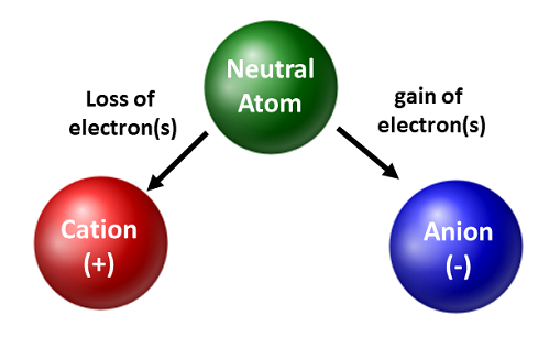
The electronic configuration determines whether an atom is more likely to lose or gain electrons:
- Atoms with few outer electrons tend to lose electrons (forming positive ions).
- Atoms with nearly full outer shells tend to gain electrons (forming negative ions).
Stability and Noble Gas Configuration
Atoms are most stable when their outer shell is full. This is why noble gases are very unreactive.
Other elements react in ways that allow them to achieve a similar full outer shell.
This concept explains many chemical behaviours, including bonding patterns and reactivity trends across the periodic table.
Summary
- Chemical properties are determined mainly by outer-shell electrons
- Elements in the same group have similar chemical properties
- Atoms react to achieve a stable (full outer shell) configuration
- Electronic configuration determines ion formation and bonding type
Example 1:
Explain why sodium and potassium have similar chemical properties.
▶️ Answer/Explanation
Both sodium and potassium are in Group 1 and have one electron in their outer shell.
They both lose this electron to form \( +1 \) ions.
Since they undergo similar reactions and form similar ions, they have similar chemical properties.
Example 2:
Explain why chlorine is more reactive than neon.
▶️ Answer/Explanation
Chlorine has seven electrons in its outer shell and needs one more electron to achieve a full outer shell.
Therefore, it is highly reactive and readily gains an electron.
Neon already has a full outer shell, making it very stable and unreactive.
Therefore, chlorine is more reactive than neon.
