Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.16 Periodic table blocks (s, p, d)-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.16 Periodic table blocks (s, p, d)- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.16 Periodic table blocks (s, p, d)- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.16 know that the Periodic Table is divided into blocks, such as s, p and d, and know the number of electrons that can occupy s, p and d sub-shells in the first four quantum shells
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.16 Periodic Table Blocks and Sub-shell Electron Capacity
The Periodic Table is arranged based on the electronic configurations of elements. One important way of organising elements is into blocks, which correspond to the type of sub-shell being filled by the outermost electrons.
The main blocks are the s-block, p-block, and d-block. Each block reflects the sub-shell into which the last electron of an atom is placed.
Blocks of the Periodic Table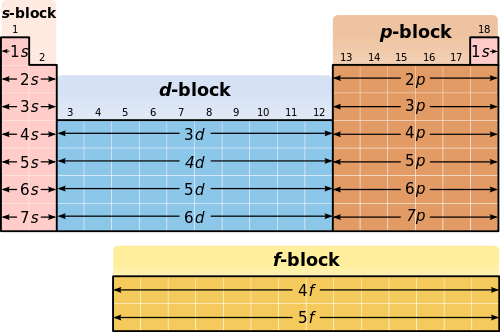
The s-block consists of elements where the outermost electrons occupy s orbitals.
These are found on the left side of the Periodic Table (Groups 1 and 2, plus helium).
The p-block consists of elements where the outermost electrons occupy p orbitals.
These are found on the right side of the Periodic Table (Groups 13 to 18).
The d-block consists of elements where electrons are being added to d orbitals.
These are the transition metals located in the centre of the Periodic Table.
The block structure arises directly from the order in which sub-shells are filled.
Sub-shells and Number of Orbitals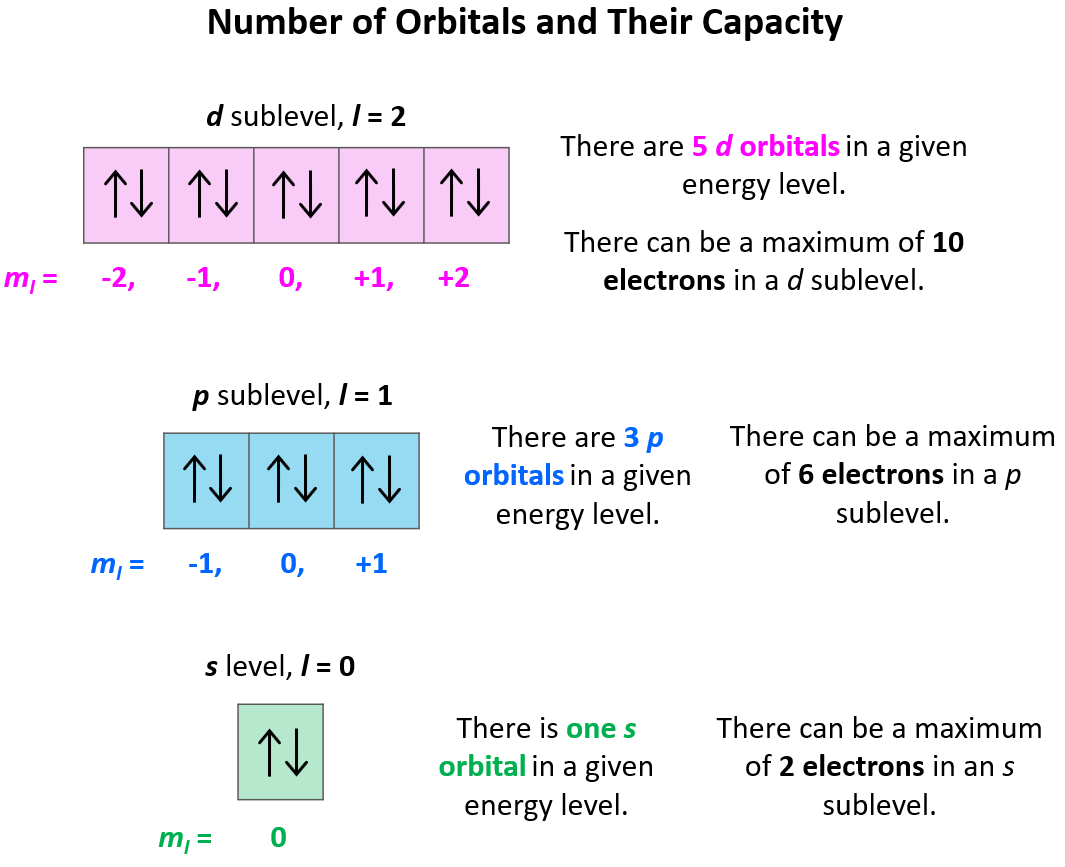
Each sub-shell consists of a specific number of orbitals:
- s sub-shell → 1 orbital
- p sub-shell → 3 orbitals
- d sub-shell → 5 orbitals
Since each orbital can hold a maximum of two electrons, the maximum number of electrons in each sub-shell is:
- s → 2 electrons
- p → 6 electrons
- d → 10 electrons
This explains why:
The s-block has 2 columns, the p-block has 6 columns, and the d-block has 10 columns in the Periodic Table.
Sub-shells in the First Four Quantum Shells
Electrons are arranged in shells (energy levels), and each shell contains specific sub-shells:
- First shell (n = 1): 1s
- Second shell (n = 2): 2s, 2p
- Third shell (n = 3): 3s, 3p, 3d
- Fourth shell (n = 4): 4s, 4p, 4d
The maximum number of electrons that can occupy each shell is determined by the sub-shells present:
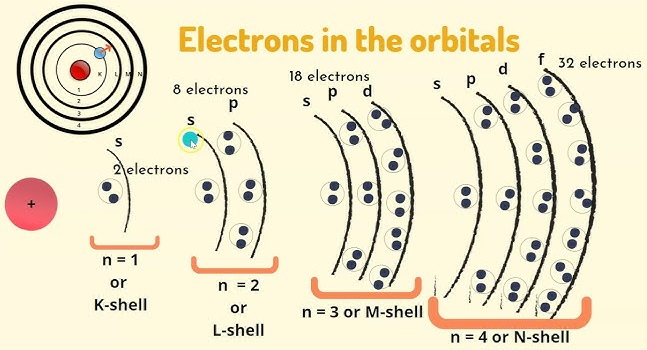
- 1st shell: 2 electrons (1s²)
- 2nd shell: 8 electrons (2s² 2p⁶)
- 3rd shell: 18 electrons (3s² 3p⁶ 3d¹⁰)
- 4th shell: 32 electrons (4s² 4p⁶ 4d¹⁰ 4f¹⁴, though f is beyond this spec)
However, for elements up to krypton, only s, p, and d sub-shells are required.
Link Between Electronic Structure and Periodic Table Layout
The structure of the Periodic Table reflects how electrons fill sub-shells:
- The period number corresponds to the highest occupied shell.
- The block indicates the type of sub-shell being filled.
- The number of columns in each block matches the maximum number of electrons that sub-shell can hold.
This shows that the Periodic Table is a direct representation of electronic structure.
Example 1:
Explain why the d-block of the Periodic Table contains 10 elements in each period.
▶️ Answer/Explanation
The d sub-shell contains 5 orbitals.
Each orbital can hold 2 electrons.
Therefore, the d sub-shell can hold a total of 10 electrons.
As each element adds one electron to the d sub-shell, there are 10 elements in the d-block.
Example 2:
Explain why the second period of the Periodic Table contains only 8 elements.
▶️ Answer/Explanation
The second shell contains the 2s and 2p sub-shells.
The 2s sub-shell can hold 2 electrons, and the 2p sub-shell can hold 6 electrons.
Therefore, the second shell can hold a total of 8 electrons.
Since each element adds one electron, there are 8 elements in the second period.
