Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.18 Period 2 & 3 trends-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.18 Period 2 & 3 trends- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.18 Period 2 & 3 trends- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.18 be able to explain:
i the trends in melting and boiling temperatures of the elements of Periods 2 and 3 of the Periodic Table in terms of the structure of the element and the bonding between its atoms or molecules
ii the general increase and the specific trends in ionisation energy of the elements across Periods 2 and 3 of the Periodic Table
iii the decrease in first ionisation energy down a group
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.18 Periodic Trends: Melting/Boiling Points and Ionisation Energy
The physical and chemical properties of elements vary in a predictable way across periods and down groups in the Periodic Table. These trends can be explained using structure, bonding, and electronic configuration.
(i) Trends in Melting and Boiling Temperatures across Periods 2 and 3
The melting and boiling temperatures of elements across Periods 2 and 3 show a distinct pattern due to changes in structure and bonding.
At the start of the period (Group 1 and 2), elements have metallic bonding.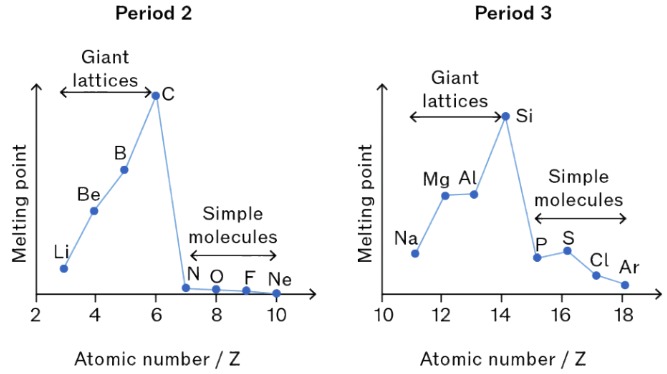
- Metal atoms form a lattice of positive ions surrounded by delocalised electrons.
- Across the period, the number of delocalised electrons increases and the positive charge of the ions increases.
- This strengthens metallic bonding, so melting and boiling points increase from Group 1 to Group 3.
In the middle of the period (e.g. carbon in Period 2, silicon in Period 3), elements form giant covalent structures.
- These structures consist of a large network of strong covalent bonds.
- A large amount of energy is required to break these bonds, so melting and boiling points are very high.
Towards the end of the period (Groups 5–7), elements exist as simple molecular substances.
- These molecules are held together by weak intermolecular forces (van der Waals forces).
- Less energy is required to overcome these forces, so melting and boiling points decrease.
Noble gases (Group 18) are monatomic and have very weak intermolecular forces, so they have the lowest melting and boiling points.
(ii) Trends in Ionisation Energy across Periods 2 and 3
There is a general increase in first ionisation energy across a period.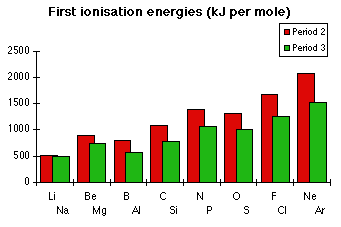
This is mainly due to:
- Increasing nuclear charge (more protons), which strengthens attraction between the nucleus and the outer electron.
- Electrons are added to the same shell, so shielding does not increase significantly.
As a result, the outer electron is held more strongly and more energy is required to remove it.
However, there are important anomalies in this trend:
Sub-shell effect (Group 2 → Group 3)
- A decrease occurs when moving from an s sub-shell to a p sub-shell (e.g. Be → B or Mg → Al).
- p electrons are higher in energy and further from the nucleus, so they are easier to remove.
Electron pairing effect (Group 5 → Group 6)
- A decrease occurs when electrons begin to pair in the same orbital (e.g. N → O or P → S).
- Repulsion between paired electrons makes it easier to remove one of them.
These irregularities provide evidence for sub-shells and electron interactions.
(iii) Decrease in Ionisation Energy down a Group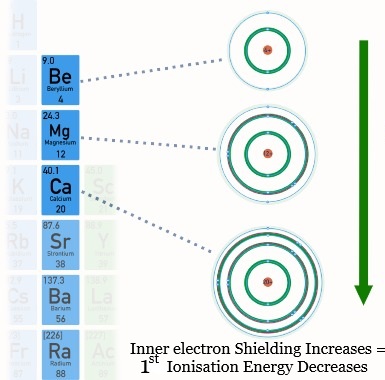
As you move down a group, the first ionisation energy decreases.
This occurs despite an increase in nuclear charge, due to two key factors:
- Increased distance: outer electrons are further from the nucleus due to additional electron shells.
- Increased shielding: inner-shell electrons reduce the attraction between the nucleus and the outer electron.
These effects outweigh the increase in nuclear charge, so less energy is required to remove the outer electron.
Therefore, ionisation energy decreases down the group.
Example 1:
Explain the trend in melting points across Period 3 from sodium to argon.
▶️ Answer/Explanation
From sodium to aluminium, melting points increase due to stronger metallic bonding caused by increasing charge and number of delocalised electrons.
Silicon has a very high melting point because it forms a giant covalent structure.
After silicon, melting points decrease sharply as elements form simple molecular structures with weak intermolecular forces.
Argon has the lowest melting point because it is monatomic with very weak forces between atoms.
Example 2:
Explain why the first ionisation energy decreases from nitrogen to oxygen, but increases overall across the period.
▶️ Answer/Explanation
Across the period, ionisation energy generally increases due to increasing nuclear charge.
However, nitrogen has three unpaired electrons in separate p orbitals, which is relatively stable.
In oxygen, one orbital contains a pair of electrons, causing repulsion.
This repulsion makes it easier to remove an electron, so oxygen has a lower ionisation energy than nitrogen.
