Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.3–2.4 Atomic number, mass number and ions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.3–2.4 Atomic number, mass number and ions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.3–2.4 Atomic number, mass number and ions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.3 know what is meant by the terms ‘atomic (proton) number’ and ‘mass number’
2.4 be able to use the atomic number and the mass number to determine the number of each type of subatomic particle in an atom or ion
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.3 Atomic (Proton) Number and Mass Number
Each element is defined by the number of protons present in the nucleus of its atoms. Two important numbers are used to describe the composition of an atom: the atomic (proton) number and the mass number.
Atomic (Proton) Number
The atomic number (also called the proton number) is the number of protons in the nucleus of an atom.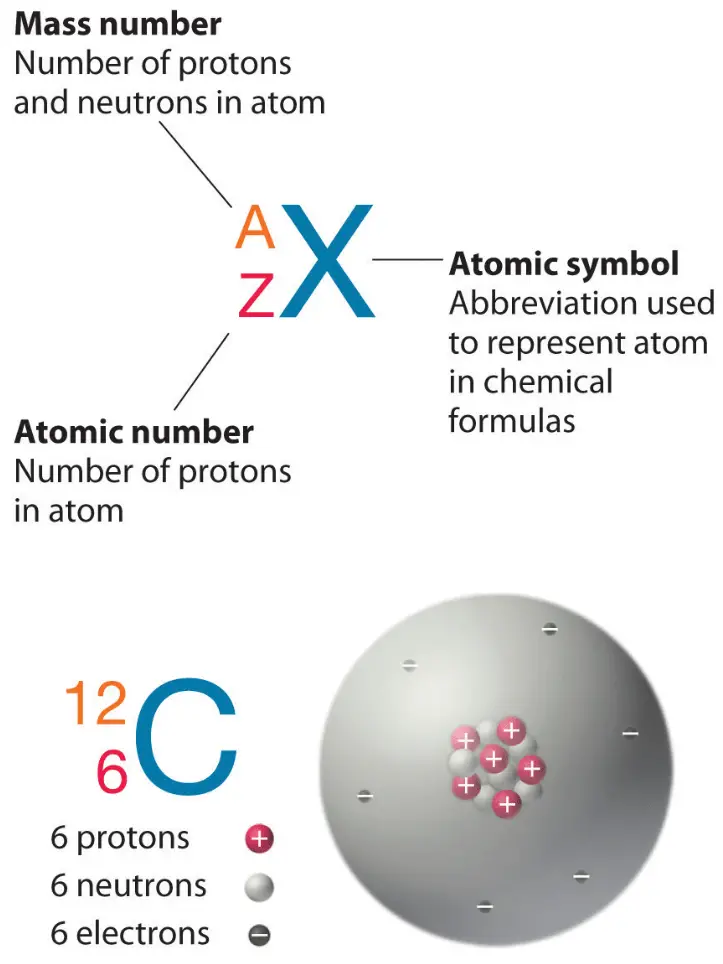
The atomic number determines the identity of an element. All atoms of the same element have the same atomic number.
In a neutral atom, the number of electrons is equal to the number of protons.
Examples
- Hydrogen has atomic number \(1\), so it contains 1 proton.
- Carbon has atomic number \(6\), so it contains 6 protons.
- Sodium has atomic number \(11\), so it contains 11 protons.
Mass Number
The mass number is the total number of protons and neutrons in the nucleus of an atom.
Since electrons have a very small mass, they are not included when calculating the mass number.
The relationship can be written as:
Mass number = number of protons + number of neutrons
\( A = Z + N \)
- \(A\) = mass number
- \(Z\) = atomic number (protons)
- \(N\) = number of neutrons
Nuclear Notation
Atoms are often written using nuclear notation:
\( ^A_Z X \)
Where:
- \(A\) = mass number
- \(Z\) = atomic number
- \(X\) = chemical symbol
Example:
\( ^{23}_{11}\mathrm{Na} \)
This shows that sodium has:
- 11 protons
- 23 − 11 = 12 neutrons
Example :
An atom of chlorine is represented as \( ^{35}_{17}\mathrm{Cl} \).
Determine:
(a) number of protons (b) number of neutrons (c) number of electrons in a neutral atom
▶️ Answer/Explanation
Atomic number \(Z = 17\)
(a) Protons = 17
(b) Neutrons:
\( N = A – Z = 35 – 17 = 18 \)
(c) In a neutral atom:
Electrons = protons = 17
Example :
An atom contains 20 neutrons and has a mass number of 39.
(a) Determine the atomic number of the atom. (b) Identify the element if the atomic number corresponds to potassium. (c) Determine the number of electrons in a neutral atom.
▶️ Answer/Explanation
Use the relationship:
\( A = Z + N \)
Substitute the values:
\( 39 = Z + 20 \)
\( Z = 19 \)
(a) Atomic number = 19
(b) Atomic number 19 corresponds to potassium \( \mathrm{K} \).
(c) In a neutral atom:
Electrons = protons = 19
2.4 Determining the Number of Subatomic Particles
The atomic number and mass number provide important information about the composition of an atom. Using these values, it is possible to determine the number of protons, neutrons, and electrons present in an atom or ion.
Number of Protons
The number of protons in an atom is equal to the atomic number.
The atomic number therefore determines the identity of the element.
Number of Neutrons
The number of neutrons is found by subtracting the atomic number from the mass number.
Number of neutrons = mass number − atomic number
\( N = A – Z \)
Number of Electrons in Atoms
In a neutral atom, the number of electrons is equal to the number of protons.
Number of electrons = atomic number
Number of Electrons in Ions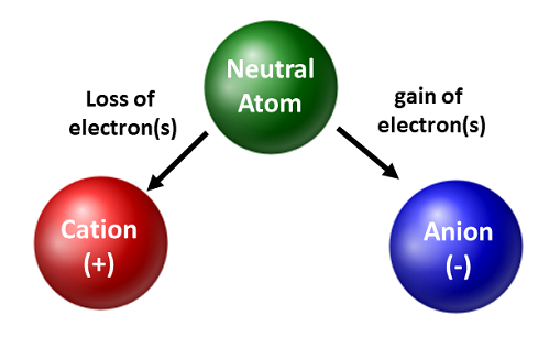
Ions are formed when atoms gain or lose electrons.
- A positive ion (cation) forms when electrons are lost.
- A negative ion (anion) forms when electrons are gained.
The number of electrons in an ion must therefore be adjusted according to the charge of the ion.
Example of Nuclear Notation
\( ^A_Z X \)
Where:
- \(A\) = mass number
- \(Z\) = atomic number
- \(X\) = chemical symbol
Example :
An atom of magnesium is represented as \( ^{24}_{12}\mathrm{Mg} \).
Determine the number of:
(a) protons (b) neutrons (c) electrons in the neutral atom
▶️ Answer/Explanation
Atomic number \( Z = 12 \)
(a) Protons = 12
(b) Neutrons:
\( N = A – Z = 24 – 12 = 12 \)
(c) Neutral atom:
Electrons = protons = 12
Example :
The ion \( ^{27}_{13}\mathrm{Al^{3+}} \) is formed from aluminium.
Determine the number of:
(a) protons (b) neutrons (c) electrons in the ion
▶️ Answer/Explanation
Atomic number \( Z = 13 \)
(a) Protons = 13
(b) Neutrons:
\( N = A – Z = 27 – 13 = 14 \)
(c) The ion has a \( 3+ \) charge, meaning it has lost three electrons.
Electrons = \( 13 – 3 = 10 \)
Therefore the ion contains 13 protons, 14 neutrons, and 10 electrons.
