Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.5 Isotopes-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.5 Isotopes- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.5 Isotopes- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.5 understand the term ‘isotope’
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.5 Isotopes
Atoms of the same element always have the same number of protons, but they may differ in the number of neutrons present in the nucleus. These different forms of the same element are called isotopes.
Isotope
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
Because isotopes have the same number of protons, they have the same atomic number and belong to the same element. However, the different numbers of neutrons result in different mass numbers.
Key Characteristics of Isotopes
- They have the same atomic number.
- They have different mass numbers.
- They have the same chemical properties because they have the same electron arrangement.
- They may have slightly different physical properties due to their different masses.
Example
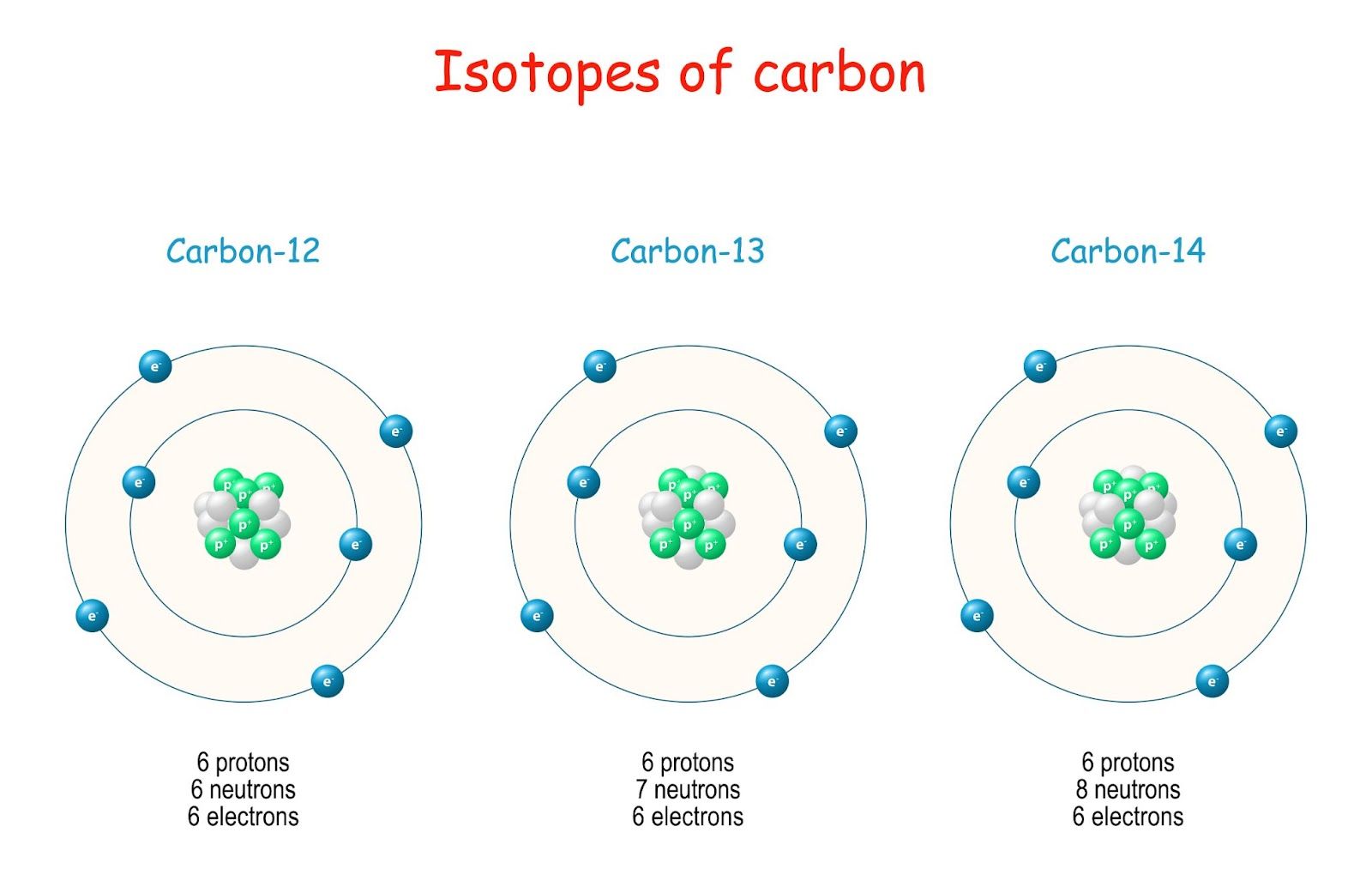
\( ^{12}_{6}\mathrm{C} \), \( ^{13}_{6}\mathrm{C} \), \( ^{14}_{6}\mathrm{C} \)
All three isotopes contain 6 protons, but they contain different numbers of neutrons:
- Carbon-12 → 6 neutrons
- Carbon-13 → 7 neutrons
- Carbon-14 → 8 neutrons
Example :
Two atoms of chlorine are represented as \( ^{35}_{17}\mathrm{Cl} \) and \( ^{37}_{17}\mathrm{Cl} \).
(a) Explain why these atoms are isotopes. (b) Determine the number of neutrons in each isotope.
▶️ Answer/Explanation
(a)
Both atoms have the same atomic number \(17\), meaning they contain the same number of protons. Therefore they are atoms of the same element.
However, they have different mass numbers (35 and 37), meaning they contain different numbers of neutrons.
This satisfies the definition of isotopes.
(b)
Neutrons in \( ^{35}\mathrm{Cl} \) \( N = 35 – 17 = 18 \)
Neutrons in \( ^{37}\mathrm{Cl} \) \( N = 37 – 17 = 20 \)
Example :
An element has two isotopes:
\( ^{63}_{29}\mathrm{X} \) and \( ^{65}_{29}\mathrm{X} \)
(a) Determine the number of protons and neutrons in each isotope. (b) Explain why these atoms have identical chemical properties.
▶️ Answer/Explanation
(a)
Atomic number = 29
Protons = 29
Neutrons in \( ^{63}\mathrm{X} \): \( 63 – 29 = 34 \)
Neutrons in \( ^{65}\mathrm{X} \): \( 65 – 29 = 36 \)
(b)
Both isotopes contain the same number of protons and therefore the same number of electrons in a neutral atom.
Since chemical reactions depend on electron arrangement, both isotopes have identical chemical properties.
