Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 2.8-2.9 I, II ,III Ionisation energy-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.8-2.9 I, II ,III Ionisation energy- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 2.8-2.9 I, II ,III Ionisation energy- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
2.8 be able to define first, second and third ionisation energies and understand that all ionisation energies are endothermic
2.9 know that an orbital is a region within an atom that can hold up to two electrons with opposite spins
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
2.8 Ionisation Energies
Ionisation energy is the energy required to remove electrons from atoms in the gaseous state. Successive ionisation energies refer to the removal of electrons one after another.
First Ionisation Energy
The first ionisation energy is the energy required to remove one electron from each atom in one mole of gaseous atoms to form one mole of gaseous 1+ ions.
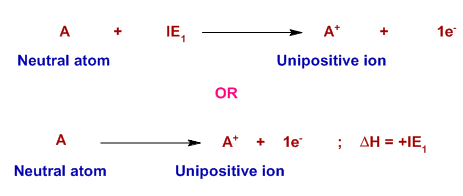
\( \mathrm{X(g) \rightarrow X^+(g) + e^-} \)
Second Ionisation Energy
The second ionisation energy is the energy required to remove one electron from each ion in one mole of gaseous 1+ ions to form one mole of gaseous 2+ ions.
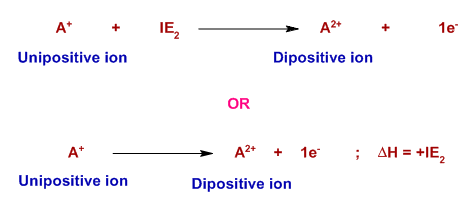
\( \mathrm{X^+(g) \rightarrow X^{2+}(g) + e^-} \)
Third Ionisation Energy
The third ionisation energy is the energy required to remove one electron from each ion in one mole of gaseous 2+ ions to form one mole of gaseous 3+ ions.
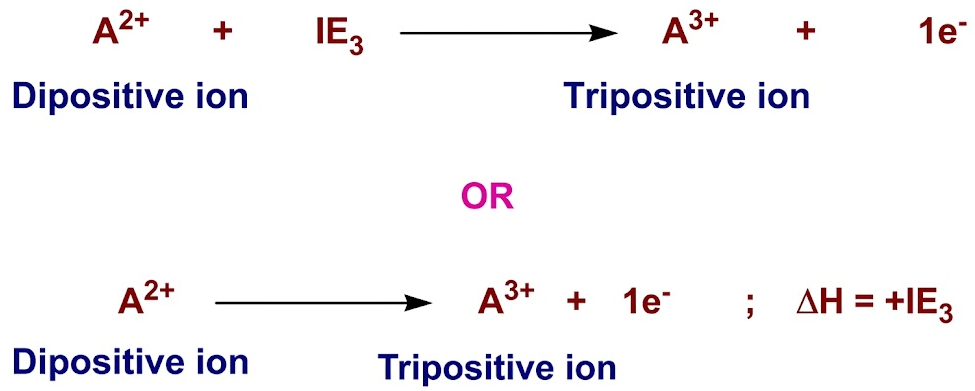
\( \mathrm{X^{2+}(g) \rightarrow X^{3+}(g) + e^-} \)
Key Points
- Ionisation energies are always measured in kJ mol\(^{-1}\).
- Each successive ionisation energy is greater than the previous one.
- This is because electrons are removed from increasingly positive ions, so the attraction between the nucleus and the remaining electrons increases.
Endothermic Nature
All ionisation energies are endothermic, meaning energy must be supplied to remove an electron from an atom or ion.
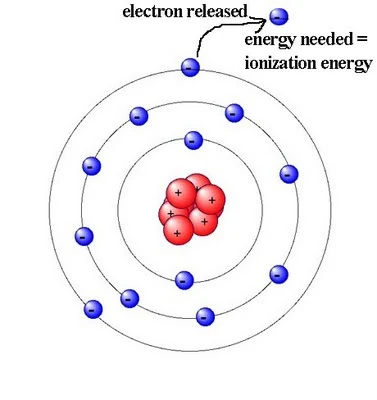
This is because energy is required to overcome the electrostatic attraction between the positively charged nucleus and the negatively charged electron.
Example 1:
The first three ionisation energies of an element are:
IE₁ = 738 kJ mol\(^{-1}\) IE₂ = 1450 kJ mol\(^{-1}\) IE₃ = 7730 kJ mol\(^{-1}\)
Explain why there is a large increase between the second and third ionisation energies.
▶️ Answer/Explanation
The large increase indicates that the third electron is removed from an inner shell.
The first two electrons are removed from the outer shell, but after these are removed, the atom has a stable electron configuration.
Removing the third electron requires much more energy because it is closer to the nucleus and more strongly attracted.
Example 2:
Write equations for the first, second, and third ionisation energies of aluminium.
▶️ Answer/Explanation
First ionisation energy
\( \mathrm{Al(g) \rightarrow Al^+(g) + e^-} \)
Second ionisation energy
\( \mathrm{Al^+(g) \rightarrow Al^{2+}(g) + e^-} \)
Third ionisation energy
\( \mathrm{Al^{2+}(g) \rightarrow Al^{3+}(g) + e^-} \)
Each process requires energy input, so all are endothermic.
2.9 Orbitals
Electrons in atoms do not move in fixed circular paths. Instead, they occupy regions of space where there is a high probability of finding an electron. These regions are called orbitals.
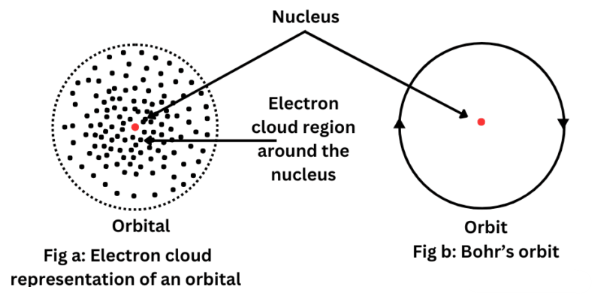
Orbital
An orbital is a region within an atom that can hold up to two electrons with opposite spins.
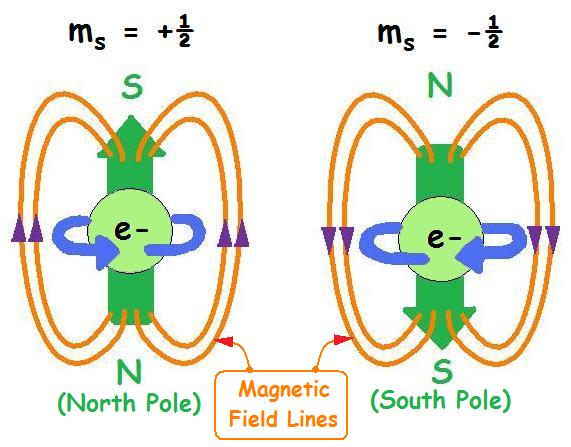
Electrons in the same orbital must have opposite spins, meaning they rotate in opposite directions. This is known as the Pauli Exclusion Principle.
Key Points
- Each orbital can hold a maximum of two electrons.
- The two electrons must have opposite spins.
- Orbitals are regions of probability, not fixed paths.
- Orbitals are arranged in shells and sublevels (e.g. s, p).
Electron Representation
Electrons in orbitals are often represented using arrows:
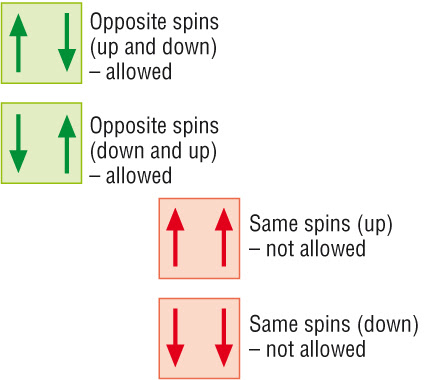
\( \uparrow \) and \( \downarrow \)
These arrows show electrons with opposite spins occupying the same orbital.
Example 1:
Explain why the following electron arrangement in a single orbital is not possible:
\( \uparrow \uparrow \)
▶️ Answer/Explanation
This arrangement is not possible because both electrons have the same spin.
According to the Pauli Exclusion Principle, two electrons in the same orbital must have opposite spins.
Therefore, a valid arrangement would be:
\( \uparrow \downarrow \)
Example 2:
An atom has the electron configuration:
\( 1s^2 \; 2s^2 \; 2p^2 \)
(a) How many orbitals are occupied in the 2p sublevel? (b) Explain how the electrons are arranged in these orbitals.
▶️ Answer/Explanation
(a)
The 2p sublevel contains three orbitals.
With 2 electrons, two orbitals are occupied.
(b)
According to Hund’s rule, electrons occupy separate orbitals first before pairing.
So the electrons are arranged as:
\( \uparrow \; \uparrow \; \_ \)
Each electron occupies a different orbital with parallel spins before pairing occurs.
