Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.1–3.2 Evidence and formation of ions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.1–3.2 Evidence and formation of ions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.1–3.2 Evidence and formation of ions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.1 know and be able to interpret evidence for the existence of ions, limited to physical properties of ionic compounds, electron density maps and the migration of ions
3.2 be able to describe the formation of ions in terms of loss or gain of electrons
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.1 Evidence for the Existence of Ions
Ions are charged particles formed when atoms gain or lose electrons. Although ions cannot be seen directly, their existence is supported by strong experimental and observational evidence.
This evidence comes from:
- the physical properties of ionic compounds
- electron density maps
- the migration of ions in electric fields
(i) Physical Properties of Ionic Compounds
Ionic compounds show characteristic physical properties that provide strong evidence for the presence of ions.
High melting and boiling points
Ionic compounds have high melting and boiling points because of strong electrostatic forces of attraction between oppositely charged ions in a giant ionic lattice.
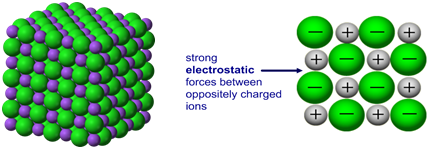
A large amount of energy is required to overcome these forces, indicating strong interactions between charged particles.
Electrical conductivity
- Solid ionic compounds do not conduct electricity because the ions are fixed in place within the lattice.
- When molten or dissolved in water, ionic compounds conduct electricity because the ions are free to move and carry charge.
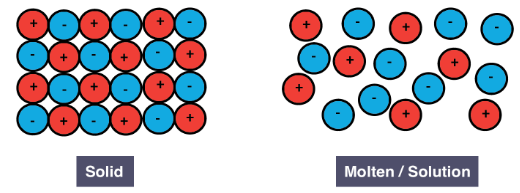
This provides clear evidence that the substance contains mobile charged particles (ions).
Solubility in water
- Many ionic compounds dissolve in water because polar water molecules surround and stabilise the ions.
- This process (hydration) separates the ions, allowing them to move freely in solution.
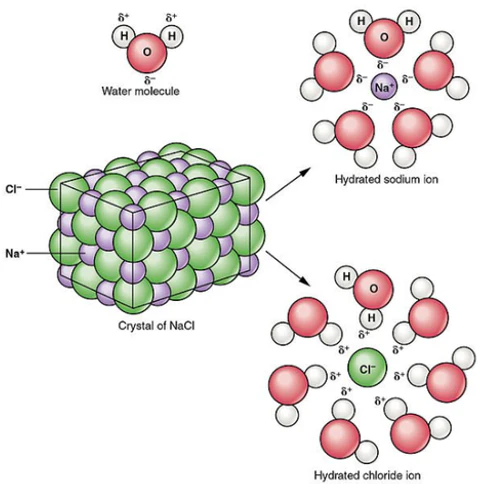
These properties together strongly support the idea that ionic compounds consist of charged particles.
(ii) Electron Density Maps
Electron density maps are obtained using techniques such as X-ray crystallography. These maps show how electron density is distributed within a structure.
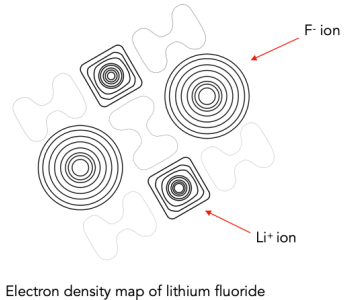
In ionic compounds, electron density is concentrated around individual ions rather than shared between atoms.
This contrasts with covalent bonding, where electron density is found between nuclei due to shared electrons.
In ionic substances:
- Cations show reduced electron density because they have lost electrons.
- Anions show increased electron density because they have gained electrons.
The clear separation of electron density around ions provides direct evidence for the existence of charged particles rather than neutral atoms.
(iii) Migration of Ions
The movement of ions in an electric field provides strong experimental evidence for their existence.
When an electric current is passed through a molten ionic compound or an aqueous solution:
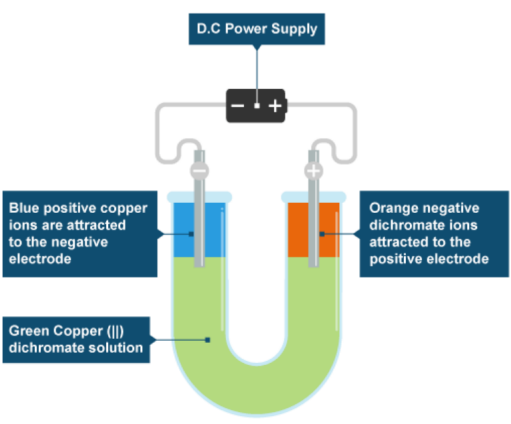
- Positive ions (cations) move towards the negative electrode (cathode).
- Negative ions (anions) move towards the positive electrode (anode).
This movement is known as electrolysis.
The fact that substances conduct electricity through the movement of charged particles provides direct evidence that ions exist and are mobile under certain conditions.
Overall Conclusion
- The physical properties of ionic compounds, electron density maps, and the movement of charged particles in electric fields all provide consistent and strong evidence for the existence of ions.
- Together, these observations confirm that ionic substances consist of positively and negatively charged particles arranged in structured lattices.
Example 1:
Explain how electrical conductivity provides evidence for the existence of ions in sodium chloride.
▶️ Answer/Explanation
Solid sodium chloride does not conduct electricity because its ions are fixed in a lattice and cannot move.
When molten or dissolved in water, it conducts electricity because the ions are free to move.
The movement of charged particles carrying current provides evidence that the substance contains ions.
Example 2:
Explain how electron density maps distinguish between ionic and covalent bonding.
▶️ Answer/Explanation
In ionic bonding, electron density is concentrated around individual ions, showing that electrons are not shared.
In covalent bonding, electron density is found between the nuclei, indicating shared electrons.
Therefore, electron density maps provide evidence for the presence of ions in ionic compounds.
3.2 Formation of Ions
Ions are formed when atoms lose or gain electrons. This process allows atoms to achieve a more stable electronic configuration, often similar to that of a noble gas.
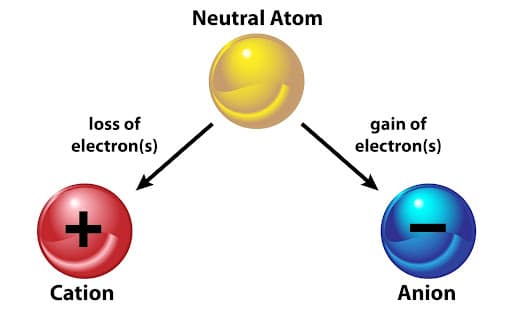
The formation of ions is driven by the tendency of atoms to reach a full outer shell, which is energetically more stable.
Formation of Positive Ions (Cations)
A cation is formed when an atom loses one or more electrons.

When electrons are lost, the number of protons exceeds the number of electrons, resulting in a net positive charge.
Metals tend to form cations because they have relatively few outer-shell electrons and low ionisation energies.
For example:
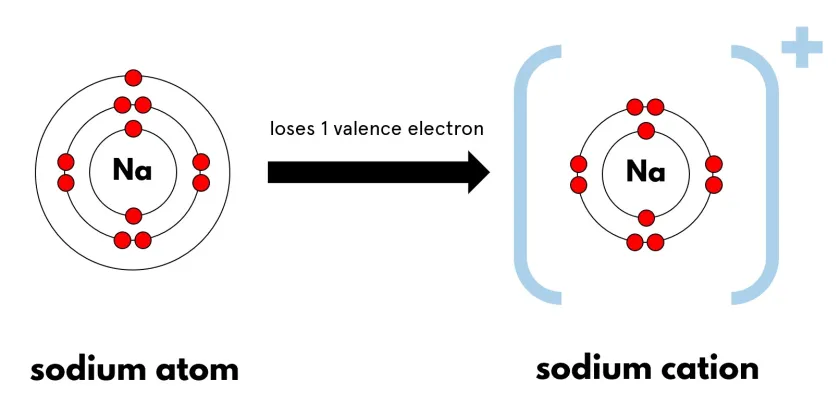
- Sodium atom: \( \mathrm{1s^2 2s^2 2p^6 3s^1} \)
- Sodium ion: \( \mathrm{1s^2 2s^2 2p^6} \)
\( \mathrm{Na \rightarrow Na^+ + e^-} \)
The sodium atom loses one electron to achieve a stable noble gas configuration.
Formation of Negative Ions (Anions)
An anion is formed when an atom gains one or more electrons.

When electrons are gained, the number of electrons exceeds the number of protons, resulting in a net negative charge.
Non-metals tend to form anions because they have nearly full outer shells and higher electron affinity.
For example:
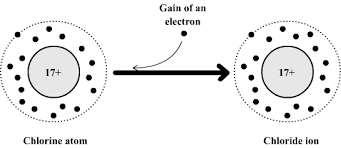
- Chlorine atom: \( \mathrm{1s^2 2s^2 2p^6 3s^2 3p^5} \)
- Chloride ion: \( \mathrm{1s^2 2s^2 2p^6 3s^2 3p^6} \)
\( \mathrm{Cl + e^- \rightarrow Cl^-} \)
The chlorine atom gains one electron to complete its outer shell.
Relationship with Group Number
The charge of ions formed by main group elements is related to their position in the Periodic Table:
- Group 1 → lose 1 electron → \( +1 \) ions
- Group 2 → lose 2 electrons → \( +2 \) ions
- Group 6 → gain 2 electrons → \( -2 \) ions
- Group 7 → gain 1 electron → \( -1 \) ions
This reflects the number of electrons needed to achieve a full outer shell.
Energy Considerations
The formation of ions involves energy changes:
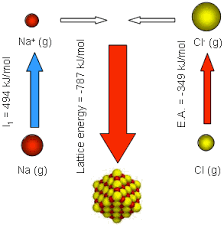
- Energy is required to remove electrons (ionisation energy).
- Energy is released when electrons are gained (electron affinity).
These energy changes influence how easily ions are formed.
Overall Concept
- Atoms form ions by losing or gaining electrons to achieve a stable electronic configuration, usually with a full outer shell.
- The type of ion formed depends on the element’s electronic configuration and its position in the Periodic Table.
Example 1:
Explain why magnesium forms a \( \mathrm{Mg^{2+}} \) ion rather than a \( \mathrm{Mg^+} \) ion.
▶️ Answer/Explanation
Magnesium has the electronic configuration \( \mathrm{2,8,2} \).
It loses two electrons to achieve a full outer shell \( \mathrm{2,8} \), which is stable.
Forming \( \mathrm{Mg^+} \) would not result in a stable configuration, so \( \mathrm{Mg^{2+}} \) is preferred.
Example 2:
Explain why oxygen forms a \( \mathrm{O^{2-}} \) ion.
▶️ Answer/Explanation
Oxygen has six electrons in its outer shell.
It gains two electrons to complete its outer shell and achieve a stable configuration.
This results in the formation of a \( \mathrm{O^{2-}} \) ion.
