Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.10 Covalent bonding -Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.10 Covalent bonding – Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.10 Covalent bonding – Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.10 understand that covalent bonding is the strong electrostatic attraction between two nuclei and the shared pair of electrons between them, based on the evidence:
i the physical properties of giant atomic structures
ii electron density maps for simple molecules
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.10 Covalent Bonding
Covalent bonding explains how atoms bond by sharing electrons, forming strong bonds within molecules or giant structures. Unlike ionic bonding, electrons are not transferred but are shared between atoms.
Covalent Bonding
Covalent bonding is the strong electrostatic attraction between two nuclei and a shared pair of electrons between them.
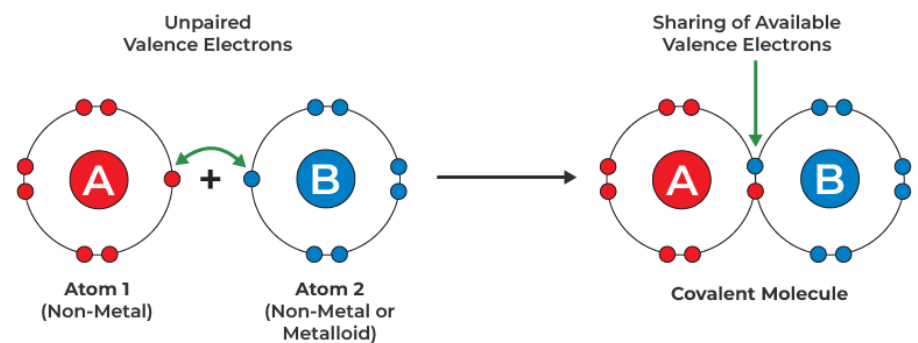
The shared pair of electrons is attracted to both nuclei simultaneously, holding the atoms together in a stable arrangement.
This type of bonding usually occurs between non-metal atoms, allowing them to achieve full outer electron shells.
Evidence for Covalent Bonding
(i) Physical Properties of Giant Atomic Structures
Giant atomic structures (giant covalent lattices) provide strong evidence for covalent bonding due to their physical properties.
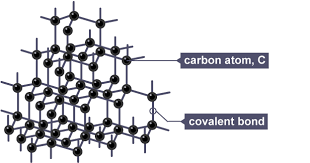
- Substances such as diamond and silicon dioxide consist of a giant network of atoms bonded by covalent bonds.
- Each atom is bonded to several others by strong covalent bonds extending throughout the structure.
- These structures have very high melting and boiling points because many strong covalent bonds must be broken.
- They are usually hard and rigid, reflecting the strength of the bonds.
- Most do not conduct electricity because they lack mobile charge carriers (except graphite).
These properties confirm that strong forces exist between atoms, consistent with covalent bonding.
(ii) Electron Density Maps
Electron density maps provide direct experimental evidence for covalent bonding.
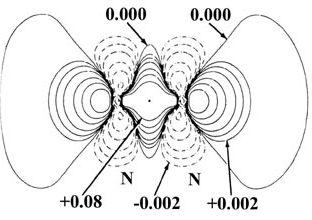
These maps show the distribution of electron density within a molecule, often obtained using techniques such as X-ray diffraction.
- Regions of high electron density are found between the nuclei of bonded atoms.
- This indicates that electrons are shared between the atoms rather than transferred.
- The shared electron pair lies in the region between the nuclei, forming a bond.
- The concentration of electron density between nuclei supports the idea of electrostatic attraction between nuclei and shared electrons.
Therefore, electron density maps provide strong experimental evidence for the existence of covalent bonds.
Summary
- Covalent bonding involves a shared pair of electrons between two atoms.
- The bond arises from electrostatic attraction between nuclei and shared electrons.
- Giant covalent structures show strong physical evidence of covalent bonding.
- Electron density maps provide direct evidence of electron sharing.
Together, these observations confirm the nature of covalent bonding.
Example 1:
Explain how the properties of diamond support the existence of covalent bonding.
▶️ Answer/Explanation
Diamond has a giant covalent structure in which each carbon atom is bonded to four others.
Strong covalent bonds extend throughout the structure.
A large amount of energy is required to break these bonds.
Therefore, diamond has a very high melting point and is extremely hard.
This supports the presence of strong covalent bonding.
Example 2:
Explain how electron density maps provide evidence for covalent bonding.
▶️ Answer/Explanation
Electron density maps show where electrons are most likely to be found.
In covalent bonds, high electron density is observed between the nuclei of bonded atoms.
This indicates that electrons are shared between the atoms.
The shared electrons attract both nuclei, forming a bond.
Therefore, these maps provide direct evidence for covalent bonding.
