Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.11 Dot-and-cross -Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.11 Dot-and-cross – Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.11 Dot-and-cross – Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.11 be able to draw dot-and-cross diagrams to show electrons in covalent substances, including:
i molecules with single, double and triple bonds
ii species with dative covalent (coordinate) bonds, including Al2_22Cl6_66 and the ammonium ion
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.11 Dot-and-Cross Diagrams
Dot-and-cross diagrams are used to represent the arrangement of electrons in covalent substances. They show how atoms share electrons to form covalent bonds and help visualise bonding clearly.
Dot-and-Cross Diagrams
Dot-and-cross diagrams show valence electrons from different atoms using different symbols (dots and crosses) to represent how electrons are shared in covalent bonds.
![]()
Each shared pair of electrons represents a covalent bond, where one electron comes from each atom (except in dative bonds).
Single, Double and Triple Bonds
Covalent bonds can involve one, two or three shared pairs of electrons.
Single bond: one shared pair of electrons.
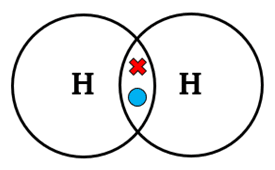
- Example: \( \mathrm{H_2} \), \( \mathrm{Cl_2} \)
Double bond: two shared pairs of electrons.
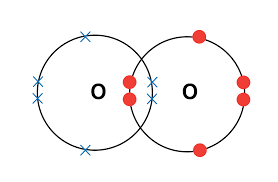
- Example: \( \mathrm{O_2} \)
Triple bond: three shared pairs of electrons.
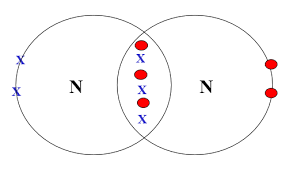
- Example: \( \mathrm{N_2} \)
Multiple bonds involve stronger attraction between nuclei and shared electrons, resulting in shorter and stronger bonds.
Dative (Coordinate) Covalent Bonds
A dative covalent bond is formed when both electrons in a shared pair are donated by one atom.

This usually occurs when one atom has a lone pair of electrons and another atom has an empty orbital.
- The donor atom provides both electrons.
- The bond is often represented by an arrow (→) showing the direction of donation.
- Once formed, the bond behaves like a normal covalent bond.
Example: Ammonium Ion (\( \mathrm{NH_4^+} \))
A lone pair on nitrogen in ammonia is donated to a hydrogen ion (\( \mathrm{H^+} \)).
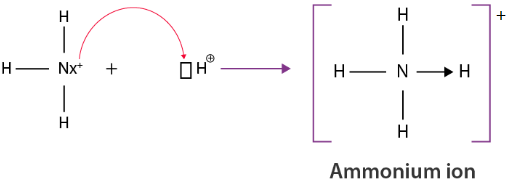
\( \mathrm{NH_3 + H^+ \rightarrow NH_4^+} \)
Nitrogen forms four bonds, one of which is a dative bond. All bonds become equivalent after formation.
Example: Aluminium Chloride Dimer (\( \mathrm{Al_2Cl_6} \))
Aluminium chloride forms a dimer where chlorine atoms donate lone pairs to aluminium atoms.
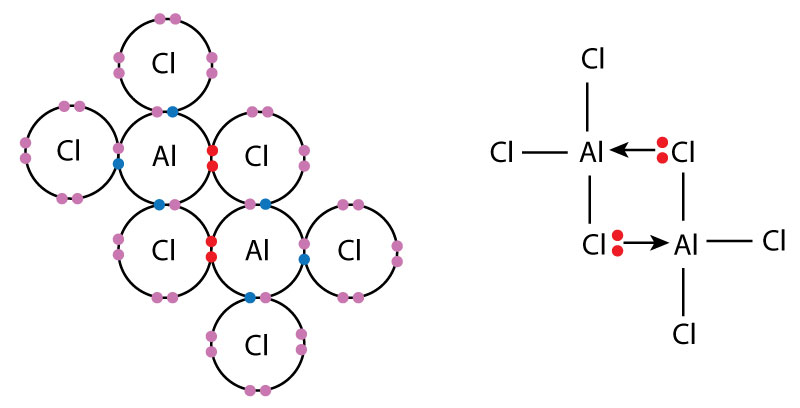
- Aluminium is electron-deficient and has an incomplete octet.
- Chlorine atoms donate lone pairs to form dative bonds.
- This results in a stable \( \mathrm{Al_2Cl_6} \) structure with bridging chlorine atoms.
Key Features of Dot-and-Cross Diagrams
- Show only outer (valence) electrons.
- Use different symbols to distinguish atoms.
- Clearly show shared pairs and lone pairs.
- Ensure atoms achieve full outer shells where possible.
Dot-and-cross diagrams are essential for visualising bonding and understanding electron sharing in covalent substances.
Example 1:
Draw and explain the dot-and-cross diagram for nitrogen gas (\( \mathrm{N_2} \)).
▶️ Answer/Explanation
Each nitrogen atom has five valence electrons.
They share three pairs of electrons to form a triple bond.
Each nitrogen also has one lone pair of electrons.
This allows both atoms to achieve a full outer shell.
The triple bond results in a strong bond between the atoms.
Example 2:
Explain the bonding in the ammonium ion using a dot-and-cross diagram.
▶️ Answer/Explanation
Nitrogen in ammonia has a lone pair of electrons.
This lone pair is donated to a hydrogen ion to form a dative bond.
The resulting ammonium ion has four N–H bonds.
One bond is formed by donation, but all bonds are equivalent.
The ion has a full outer shell and a positive charge.
