Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.12 Giant covalent structures-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.12 Giant covalent structures- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.12 Giant covalent structures- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.12 be able to describe the different structures formed by giant lattices of carbon atoms, including graphite, diamond and graphene, and discuss the applications of each
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.12 Giant Covalent Structures of Carbon
Carbon forms several different giant covalent (giant atomic) structures due to its ability to form four covalent bonds. These structures have different arrangements of atoms, leading to very different physical properties and applications.
Diamond
Diamond has a three-dimensional tetrahedral structure where each carbon atom is covalently bonded to four other carbon atoms.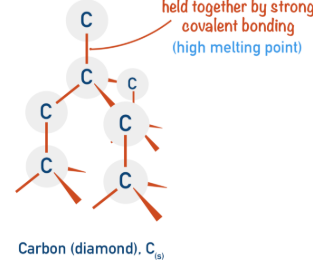
- Each carbon forms four strong covalent bonds in a rigid 3D lattice.
- There are no free electrons or ions.
- The structure extends throughout the entire crystal.
Properties
- Very high melting point due to many strong covalent bonds.
- Extremely hard because of the rigid structure.
- Does not conduct electricity as there are no mobile charge carriers.
Applications
- Used in cutting tools and drill bits due to its hardness.
- Used in jewellery due to its appearance and durability.
Graphite
Graphite consists of layers of carbon atoms arranged in hexagonal rings. Each carbon atom forms three covalent bonds.
- Each carbon atom is bonded to three others in a planar layer.
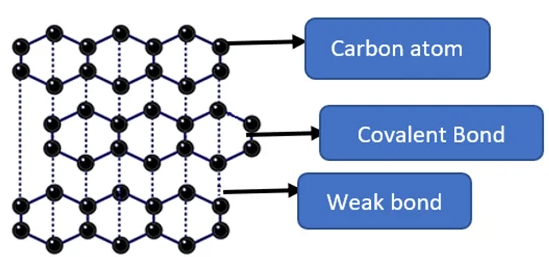
- The fourth electron is delocalised and free to move.
- Layers are held together by weak intermolecular forces.
Properties
- Conducts electricity due to delocalised electrons.
- High melting point due to strong covalent bonds within layers.
- Soft and slippery because layers can slide over each other.
Applications
- Used as a lubricant due to sliding layers.
- Used in electrodes because it conducts electricity.
- Used in pencils (graphite core).
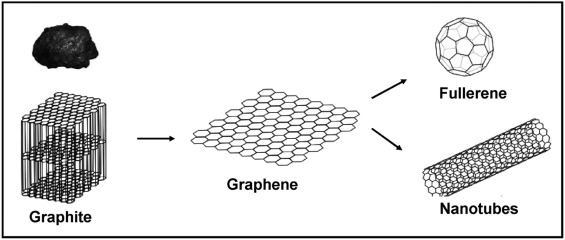
Graphene
Graphene is a single layer of carbon atoms arranged in a hexagonal lattice, essentially one layer of graphite.
- Each carbon atom is bonded to three others.
- It has delocalised electrons across the sheet.
- It is only one atom thick.
Properties
- Extremely strong despite being very thin.
- Excellent electrical conductivity.
- Very lightweight and flexible.
Applications
- Used in electronics and conductive materials.
- Used in composite materials to increase strength.
- Potential use in flexible screens and advanced technologies.
Comparison of Structures
- Diamond: 3D structure, no free electrons → very hard, non-conductive.
- Graphite: layered structure, delocalised electrons → conducts electricity, soft.
- Graphene: single layer, delocalised electrons → very strong and conductive.
The differences in bonding and structure explain the wide range of properties and applications of these carbon allotropes.
Example 1:
Explain why diamond does not conduct electricity but graphite does.
▶️ Answer/Explanation
In diamond, all four valence electrons of each carbon atom are used in covalent bonds.
There are no free electrons available to carry charge.
Therefore, diamond does not conduct electricity.
In graphite, each carbon atom uses three electrons for bonding.
The fourth electron is delocalised and free to move.
These mobile electrons carry charge, so graphite conducts electricity.
Example 2:
Explain why graphite is soft and can be used as a lubricant.
▶️ Answer/Explanation
Graphite consists of layers of carbon atoms held together by weak intermolecular forces.
These weak forces can be easily overcome.
As a result, the layers can slide over each other.
This makes graphite soft and slippery.
Therefore, it can be used as a lubricant.
