Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.13 Electronegativity-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.13 Electronegativity- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.13 Electronegativity- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.13 understand the meaning of the term ‘electronegativity’ as applied to atoms in a covalent bond
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.13 Electronegativity
In covalent bonding, atoms share electrons, but the shared pair is not always shared equally. The ability of an atom to attract the shared electrons in a covalent bond is described by its electronegativity.
Electronegativity
Electronegativity is the ability of an atom to attract the bonding pair of electrons in a covalent bond.

When two atoms form a covalent bond, both nuclei attract the shared pair of electrons. However, if one atom has a greater electronegativity, it attracts the electrons more strongly.
Effect of Electronegativity on Bonding
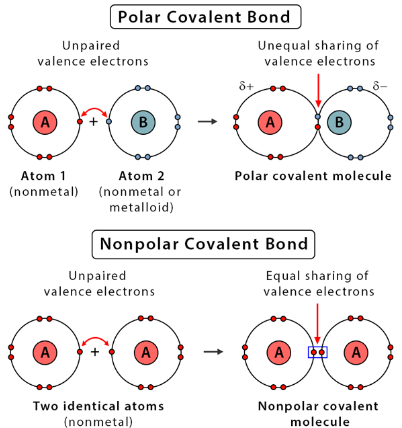
- If both atoms have the same electronegativity, the electrons are shared equally.
- This forms a non-polar covalent bond.
- If there is a difference in electronegativity, electrons are shared unequally.
- This creates a polar covalent bond.
- The more electronegative atom gains a partial negative charge (δ−), while the other gains a partial positive charge (δ+).
Therefore, electronegativity determines how electrons are distributed within a covalent bond.
Factors Affecting Electronegativity
Electronegativity depends on how strongly the nucleus attracts electrons.
- Atoms with a higher nuclear charge attract electrons more strongly.
- Smaller atomic radius increases attraction because electrons are closer to the nucleus.
- Greater shielding reduces attraction between nucleus and bonding electrons.
Trends in Electronegativity
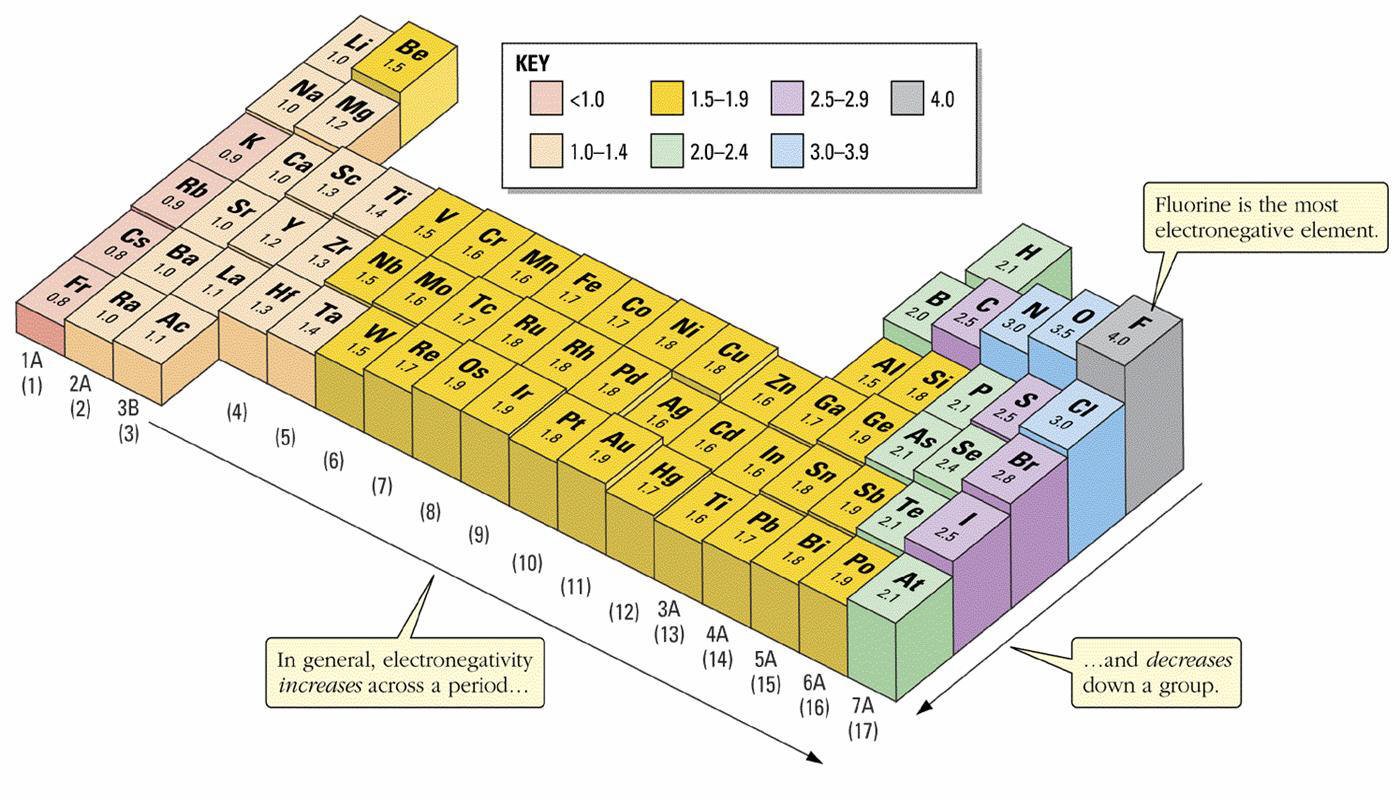
- Electronegativity increases across a period due to increasing nuclear charge.
- Electronegativity decreases down a group due to increased atomic radius and shielding.
- Fluorine is the most electronegative element.
Importance of Electronegativity
- Determines bond polarity.
- Helps explain molecular properties such as dipole moments.
- Influences the type of bonding (covalent vs ionic character).
Therefore, electronegativity is a key concept in understanding how covalent bonds behave and how electrons are distributed within molecules.
Example 1:
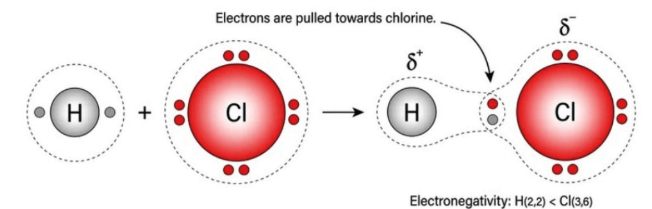
Explain why the bond in hydrogen chloride is polar.
▶️ Answer/Explanation
Chlorine is more electronegative than hydrogen.
It attracts the shared pair of electrons more strongly.
This results in an unequal distribution of electron density.
Chlorine becomes partially negative (δ−) and hydrogen becomes partially positive (δ+).
Therefore, the bond is polar.
Example 2:
Explain why the bond in chlorine gas is non-polar.
▶️ Answer/Explanation
Both chlorine atoms have the same electronegativity.
They attract the shared pair of electrons equally.
This results in equal sharing of electrons.
No partial charges are formed.
Therefore, the bond is non-polar.
