Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.14 Bonding continuum-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.14 Bonding continuum- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.14 Bonding continuum- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.14 know that ionic and covalent bonding are the extremes of a continuum of bonding type and be able to explain this in terms of electronegativity differences, leading to bond polarity in bonds and molecules, and to ionic bonding if the electronegativity is large enough
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.14 Continuum of Ionic and Covalent Bonding
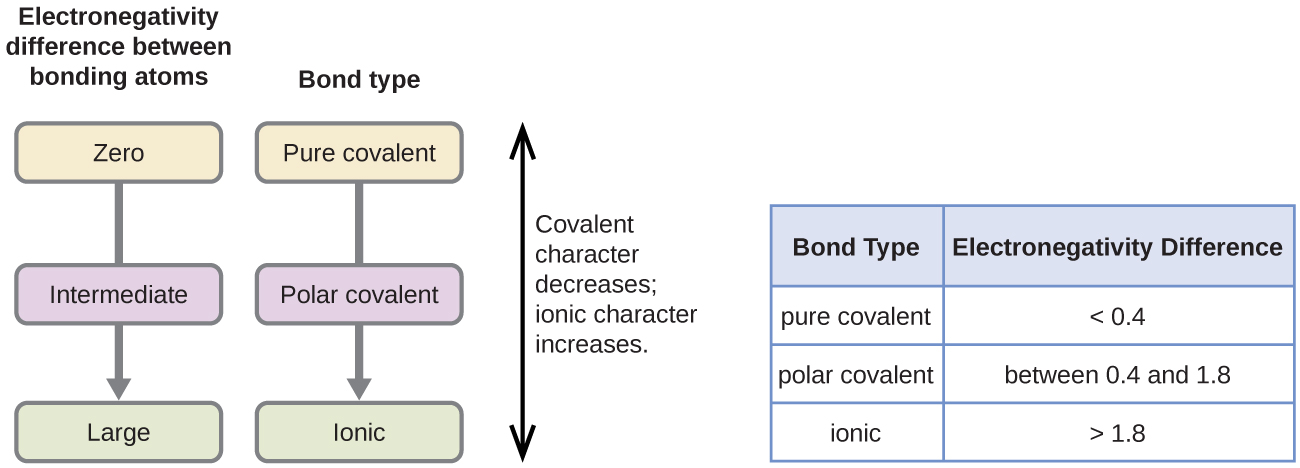
Bonding between atoms is not always purely ionic or purely covalent. Instead, bonding exists on a continuum, with ionic and covalent bonding representing two extremes. Most bonds lie somewhere in between these extremes.
Bonding Continuum
Ionic and covalent bonding are the extremes of a continuum of bonding type, depending on how electrons are distributed between atoms.
In reality, electron transfer and electron sharing are not completely separate processes. Instead, bonds can have both ionic and covalent character.
Role of Electronegativity Difference
The position of a bond on the continuum depends on the difference in electronegativity between the bonded atoms.
- If the electronegativity difference is zero or very small, electrons are shared equally.
- This results in a non-polar covalent bond.
- If the difference is moderate, electrons are shared unequally.
- This forms a polar covalent bond.
- If the difference is very large, electrons are effectively transferred.
- This results in ionic bonding.
Therefore, increasing electronegativity difference shifts bonding from covalent to ionic character.
Bond Polarity
Bond polarity arises when electrons are shared unequally due to differences in electronegativity.
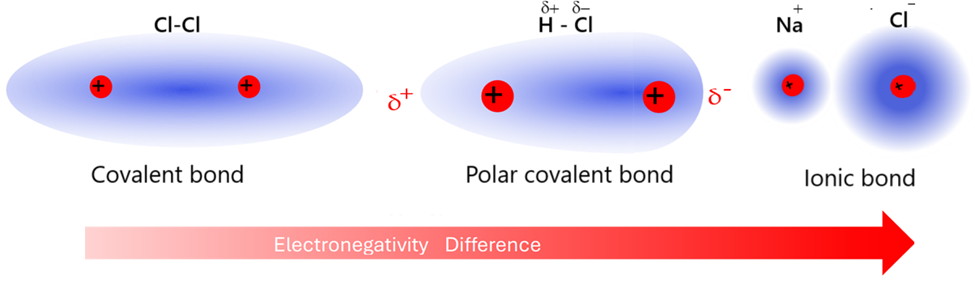
- The more electronegative atom attracts electron density more strongly.
- This creates partial charges (δ− and δ+).
- The bond becomes polar, with a dipole.
Bond polarity is an intermediate stage between non-polar covalent and ionic bonding.
Polarity in Molecules
The overall polarity of a molecule depends on both bond polarity and molecular shape.
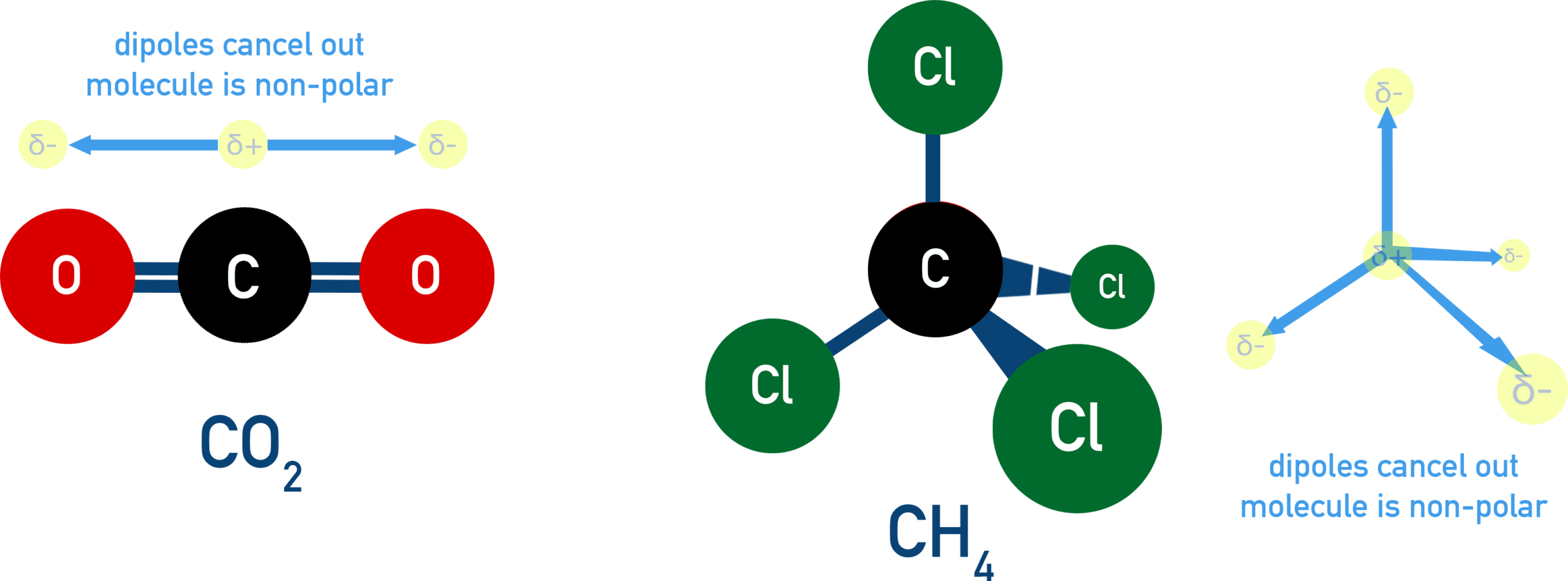
- If polar bonds are arranged symmetrically, their dipoles cancel.
- The molecule is non-polar.
- If dipoles do not cancel, the molecule is polar.
Therefore, molecular shape plays a key role in determining overall polarity.
Summary of the Continuum
- Small electronegativity difference → non-polar covalent bonding.
- Moderate difference → polar covalent bonding.
- Large difference → ionic bonding.
- Most bonds show a mixture of ionic and covalent character.
Thus, bonding should be viewed as a continuous range rather than distinct categories.
Example 1:
Explain why hydrogen chloride is considered polar covalent rather than ionic.
▶️ Answer/Explanation
There is a difference in electronegativity between hydrogen and chlorine.
Chlorine attracts the shared electrons more strongly.
This results in unequal sharing of electrons.
However, the difference is not large enough for complete electron transfer.
Therefore, the bond is polar covalent rather than ionic.
Example 2:
Explain why sodium chloride is considered ionic rather than covalent.
▶️ Answer/Explanation
There is a large electronegativity difference between sodium and chlorine.
Sodium loses an electron and chlorine gains an electron.
This results in the formation of oppositely charged ions.
Strong electrostatic attraction occurs between these ions.
Therefore, the bonding is ionic.
