Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.15 Polar bonds and polar molecules-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.15 Polar bonds and polar molecules- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.15 Polar bonds and polar molecules- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.15 be able to distinguish between polar bonds and polar molecules and predict whether or not a given molecule is likely to be polar
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.15 Polar Bonds and Polar Molecules
It is important to distinguish between polar bonds and polar molecules, as a molecule can contain polar bonds but still be non-polar overall. This depends on both electronegativity differences and the shape of the molecule.
Polar Bond
A polar bond is a covalent bond in which the bonding pair of electrons is shared unequally due to a difference in electronegativity between the atoms.
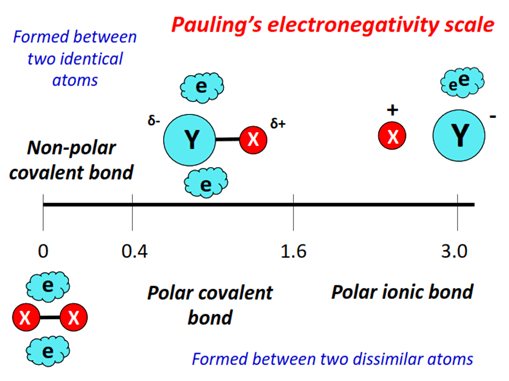
The more electronegative atom attracts the shared electrons more strongly, creating a difference in charge.
- The more electronegative atom becomes δ− (partially negative).
- The less electronegative atom becomes δ+ (partially positive).
- This creates a bond dipole.
Therefore, a polar bond depends only on the difference in electronegativity between two atoms.
Polar Molecule
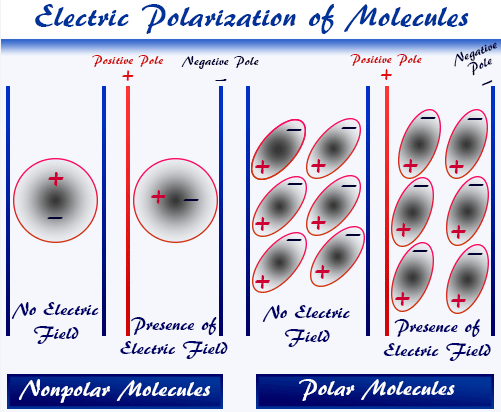
A polar molecule is a molecule that has a net dipole moment due to an uneven distribution of electron density.
Whether a molecule is polar depends on both the polarity of its bonds and its molecular shape.
- If bond dipoles cancel out due to symmetry, the molecule is non-polar.
- If bond dipoles do not cancel, the molecule is polar.
Therefore, molecular polarity depends on both electronegativity differences and geometry.
Key Difference
- A polar bond refers to a single bond between two atoms.
- A polar molecule refers to the overall molecule.
- A molecule can have polar bonds but still be non-polar overall.
Predicting Molecular Polarity
To determine whether a molecule is polar, both bond polarity and molecular shape must be considered.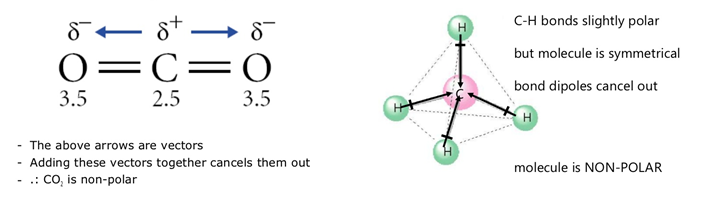
- Step 1: Identify if bonds are polar (electronegativity difference).
- Step 2: Determine the shape of the molecule.
- Step 3: Check if dipoles cancel (symmetrical) or not (asymmetrical).
Therefore, polarity is determined by the vector sum of all bond dipoles.
Examples
- \( \mathrm{HCl} \): polar bond → polar molecule (no cancellation).
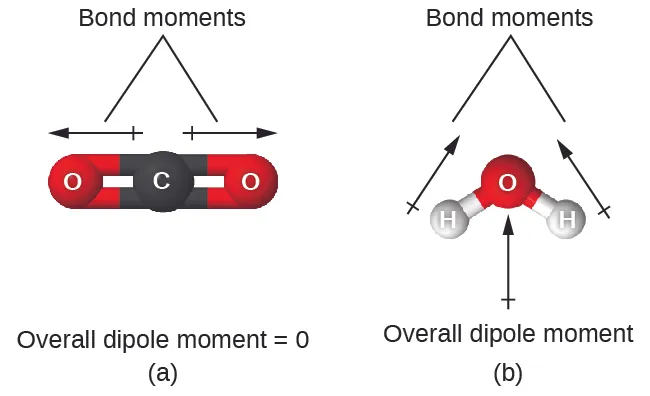
- \( \mathrm{CO_2} \): polar bonds → non-polar molecule (linear, dipoles cancel).
- \( \mathrm{H_2O} \): polar bonds → polar molecule (bent shape, dipoles do not cancel).
These examples highlight the importance of molecular shape in determining overall polarity.
Example 1:
Explain why carbon dioxide is non-polar despite having polar bonds.
▶️ Answer/Explanation
Each C=O bond is polar due to electronegativity difference.
However, the molecule is linear and symmetrical.
The bond dipoles act in opposite directions and cancel each other out.
Therefore, there is no overall dipole moment.
Hence, carbon dioxide is non-polar.
Example 2:
Explain why water is a polar molecule.
▶️ Answer/Explanation
The O–H bonds are polar due to electronegativity difference.
The molecule has a bent shape due to lone pairs on oxygen.
The bond dipoles do not cancel.
This results in a net dipole moment.
Therefore, water is a polar molecule.
Further Suggested Practical: Effect of Electrostatic Force on Liquid Jets
This practical demonstrates how molecules respond to an external electric field and can be used to determine whether a liquid is polar or non-polar. It provides experimental evidence for molecular polarity.
Principle
A polar molecule has a permanent dipole and will be attracted by an electrostatic force, causing the liquid stream to bend. Non-polar molecules do not have a permanent dipole and are much less affected.
When a charged object is brought near a liquid stream, the electric field interacts with the molecules in the liquid. The extent of deflection depends on molecular polarity.
Method
- A thin stream of liquid is allowed to flow from a burette or pipette.
- A plastic rod is charged by rubbing it with a dry cloth.
- The charged rod is brought close to the liquid stream.
- The deflection (bending) of the liquid stream is observed.
- The experiment is repeated for different liquids: water, ethanol, and cyclohexane.
Observations
- Water: stream bends strongly towards the charged rod.
- Ethanol: stream bends slightly.
- Cyclohexane: little or no deflection observed.
Explanation
- Water molecules are highly polar, so they align with the electric field and are strongly attracted.
- Ethanol is polar but less polar than water, so the deflection is smaller.
- Cyclohexane is non-polar and has no permanent dipole.
- Therefore, cyclohexane shows little or no interaction with the electric field.
Conclusion
The greater the deflection of a liquid stream, the more polar the molecules are. Polar liquids are attracted to an electrostatic field, while non-polar liquids are not significantly affected.
Evaluation and Limitations
- The strength of the charge on the rod may vary, affecting results.
- Air currents may disturb the liquid stream.
- The thickness and speed of the liquid stream may affect the degree of bending.
Despite these limitations, the experiment provides clear qualitative evidence for molecular polarity.
Example 1:
Explain why water shows a greater deflection than ethanol in this experiment.
▶️ Answer/Explanation
Water is more polar than ethanol.
It has a larger permanent dipole moment.
Therefore, it interacts more strongly with the electric field.
This results in a greater deflection of the liquid stream.
Example 2:
Explain how this experiment can be used to identify whether an unknown liquid is polar.
▶️ Answer/Explanation
The unknown liquid is tested by observing its deflection in an electric field.
If the liquid stream bends significantly, it indicates the presence of a permanent dipole.
This means the liquid is polar.
If there is little or no deflection, the liquid is likely non-polar.
Therefore, the degree of bending can be used to determine polarity.
