Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.16 Electron-pair repulsion theory-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.16 Electron-pair repulsion theory- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.16 Electron-pair repulsion theory- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.16 understand the principles of the electron-pair repulsion theory, used to interpret and predict the shapes of simple molecules and ions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.16 Electron Pair Repulsion Theory (VSEPR)
The shapes of molecules and ions can be explained using the electron-pair repulsion theory, also known as VSEPR (Valence Shell Electron Pair Repulsion) theory. This theory allows us to predict molecular geometry based on the arrangement of electron pairs around a central atom.
Principle of Electron Pair Repulsion
Electron pairs around a central atom arrange themselves as far apart as possible in order to minimise repulsion between them.
This is because electron pairs are negatively charged and repel each other. The arrangement that minimises repulsion determines the shape of the molecule or ion.
Types of Electron Pairs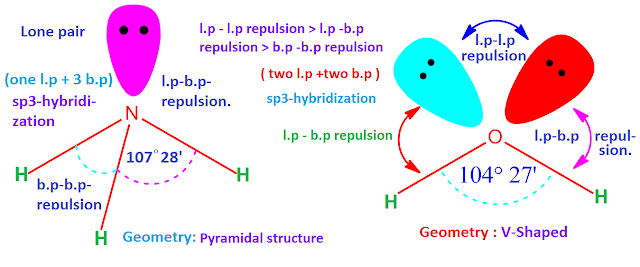
- Bonding pairs: shared between two atoms in a covalent bond.
- Lone pairs: unshared pairs of electrons on the central atom.
Lone pairs occupy more space than bonding pairs because they are only attracted to one nucleus, so they repel more strongly.
Order of Repulsion
- Lone pair – lone pair > lone pair – bonding pair > bonding pair – bonding pair
This means lone pairs have the greatest effect on bond angles and molecular shape.
Basic Shapes from Electron Pair Arrangement
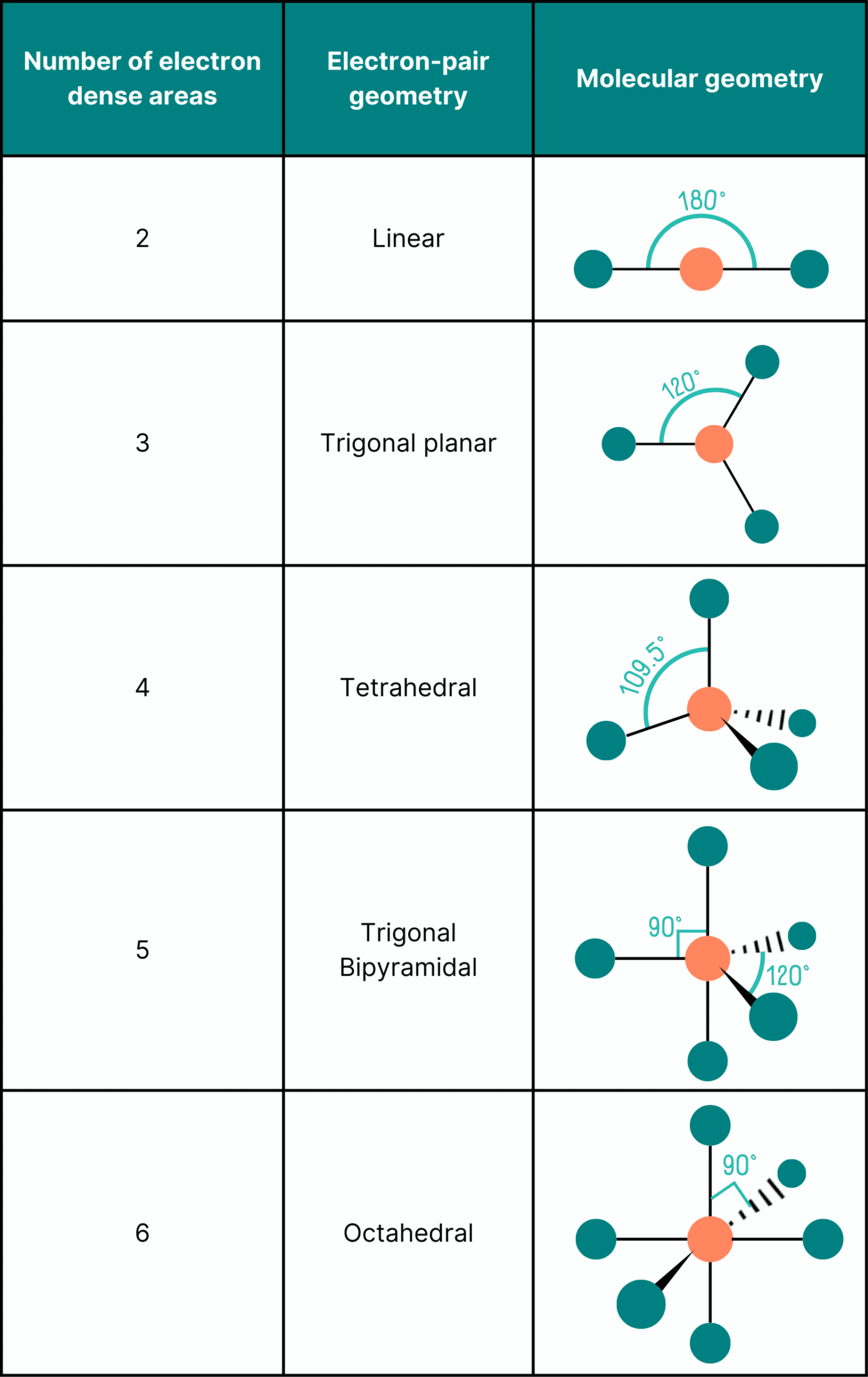
These are the shapes based on electron pair geometry, before considering lone pairs.
Effect of Lone Pairs on Shape
Lone pairs distort bond angles because they repel more strongly than bonding pairs.
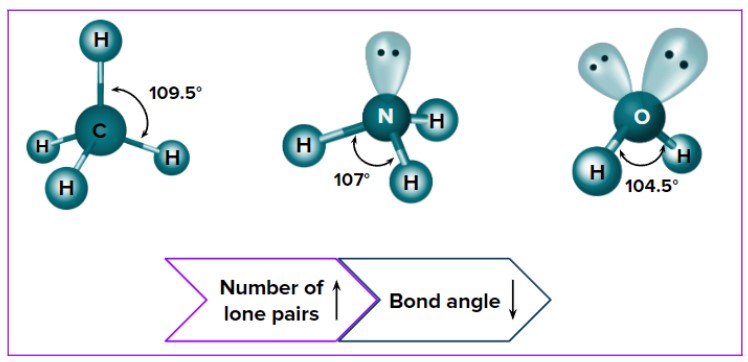
- They push bonding pairs closer together.
- This reduces bond angles from their ideal values.
- The molecular shape is determined by the positions of atoms, not lone pairs.
Examples of Molecular Shapes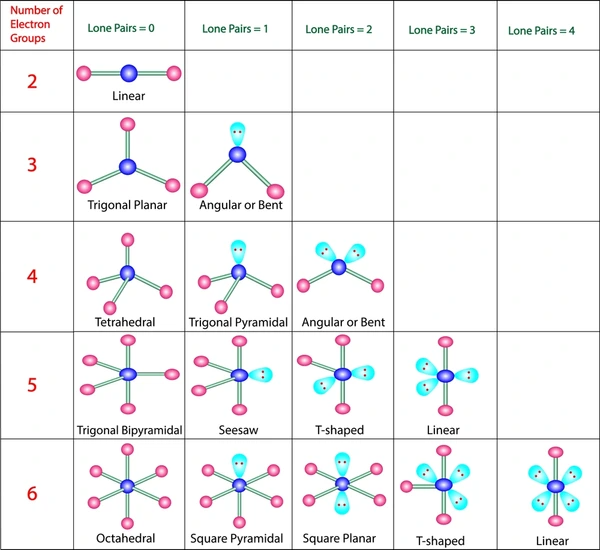
- \( \mathrm{CO_2} \): 2 bonding pairs, 0 lone pairs → linear (180°)
- \( \mathrm{BF_3} \): 3 bonding pairs, 0 lone pairs → trigonal planar (120°)
- \( \mathrm{CH_4} \): 4 bonding pairs, 0 lone pairs → tetrahedral (109.5°)
- \( \mathrm{NH_3} \): 3 bonding pairs, 1 lone pair → trigonal pyramidal (~107°)
- \( \mathrm{H_2O} \): 2 bonding pairs, 2 lone pairs → bent (~104.5°)
These examples show how lone pairs reduce bond angles and change molecular shape.
Summary
- Electron pairs repel and arrange themselves as far apart as possible.
- Lone pairs repel more strongly than bonding pairs.
- Molecular shape depends on the number of bonding pairs and lone pairs.
- Bond angles are reduced when lone pairs are present.
Therefore, electron-pair repulsion theory provides a simple and effective way to predict molecular shapes and bond angles.
Example 1:
Explain why the bond angle in ammonia is smaller than in methane.
▶️ Answer/Explanation
Methane has four bonding pairs and no lone pairs, giving a tetrahedral shape with bond angle 109.5°.
Ammonia has three bonding pairs and one lone pair.
The lone pair repels bonding pairs more strongly.
This pushes the bonding pairs closer together.
Therefore, the bond angle in ammonia is reduced to about 107°.
Example 2:
Predict and explain the shape of water using electron pair repulsion theory.
▶️ Answer/Explanation
Oxygen has four electron pairs: two bonding pairs and two lone pairs.
These arrange in a tetrahedral electron geometry.
Lone pairs repel more strongly than bonding pairs.
This pushes the bonding pairs closer together.
The molecule has a bent shape with a bond angle of about 104.5°.
