Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.17 Bond length and bond angle-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.17 Bond length and bond angle- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.17 Bond length and bond angle- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.17 understand the terms ‘bond length’ and ‘bond angle’
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
Bond Length
Bond length is the distance between the nuclei of two bonded atoms in a covalent bond.
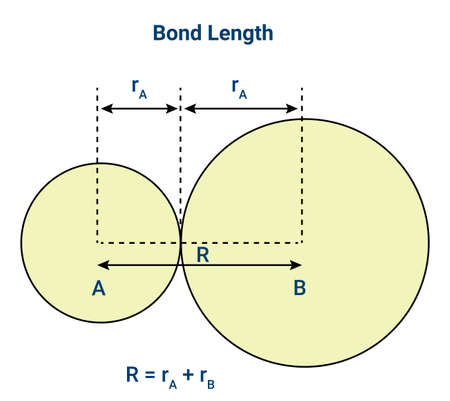
It represents the average equilibrium distance between two atoms, because atoms are constantly vibrating due to thermal energy.
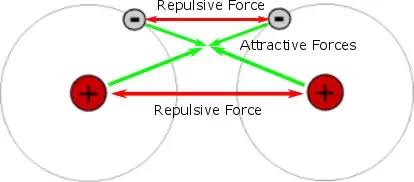
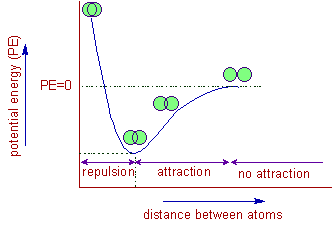
Bond length is determined by a balance between attractive forces (between nuclei and shared electrons) and repulsive forces (between nuclei and between electrons).
- When atoms are too far apart, attractive forces dominate and pull them closer.
- When atoms are too close, repulsive forces increase and push them apart.
- The bond length is the distance at which these forces are balanced.
Factors Affecting Bond Length
1. Atomic Radius
- Larger atoms have larger electron clouds, so nuclei are further apart.
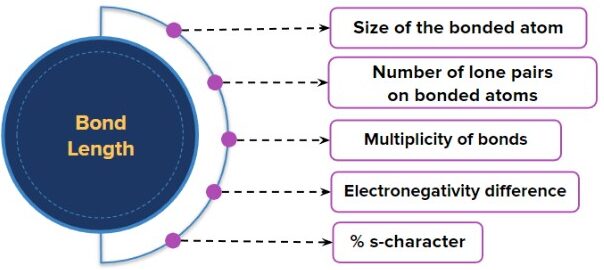
- Therefore, bonds between larger atoms have longer bond lengths.
- Example: \( \mathrm{C-C} \) bond is shorter than \( \mathrm{Si-Si} \).
2. Bond Order
- Bond order refers to the number of shared electron pairs.
- Single bond > Double bond > Triple bond (in length).
- More shared electrons increase electrostatic attraction.
- This pulls nuclei closer together, reducing bond length.
3. Bond Strength
- Stronger bonds have greater attraction between nuclei and electrons.
- This results in shorter bond lengths.
- Therefore, shorter bonds are generally stronger.
4. Electronegativity
- Greater electronegativity difference can increase attraction of electrons.
- This can slightly shorten bond length in polar bonds.
Importance of Bond Length
- Determines the size of molecules.
- Indicates bond strength.
- Helps in understanding molecular structure and reactivity.
Therefore, bond length is a key structural parameter that reflects both atomic size and the strength of bonding interactions.
Example 1:
Explain why a carbon–carbon triple bond is shorter than a carbon–carbon single bond.
▶️ Answer/Explanation
A triple bond involves three shared pairs of electrons, while a single bond has only one.
This results in much stronger electrostatic attraction between nuclei and electrons.
The increased attraction pulls the nuclei closer together.
Therefore, the bond length is shorter in a triple bond.
Example 2:
Explain why the bond length in hydrogen chloride is longer than in hydrogen fluoride.
▶️ Answer/Explanation
Chlorine atoms are larger than fluorine atoms.
This means the distance between nuclei is greater in H–Cl.
Therefore, the bond length in hydrogen chloride is longer than in hydrogen fluoride.
Bond Angle
Bond angle is the angle between two bonds that originate from the same atom.
It describes the three-dimensional arrangement of atoms in a molecule and is a key factor in determining molecular shape.
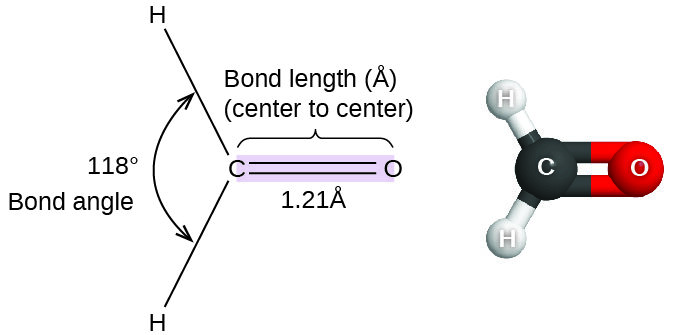
Bond angles arise due to the repulsion between electron pairs around a central atom, as explained by electron pair repulsion theory (VSEPR).
Origin of Bond Angles
Electron pairs repel each other and arrange themselves as far apart as possible to minimise repulsion.
- This leads to specific geometrical arrangements.
- Each arrangement has characteristic bond angles.
- These are called ideal bond angles.
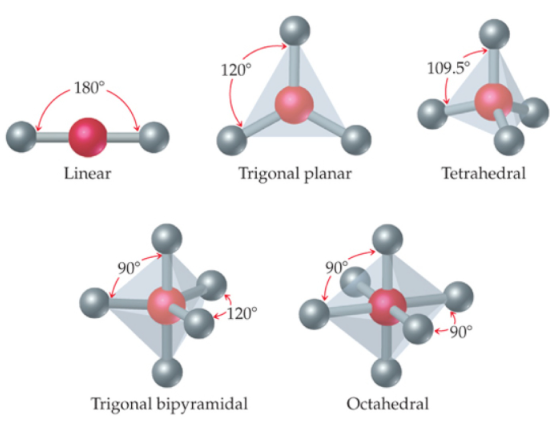
Ideal Bond Angles
- Linear → 180°
- Trigonal planar → 120°
- Tetrahedral → 109.5°
- Trigonal bipyramidal → 120° and 90°
- Octahedral → 90°
These angles occur when there are only bonding pairs and no lone pairs.
Factors Affecting Bond Angle
1. Lone Pairs
- Lone pairs repel more strongly than bonding pairs.
- This pushes bonding pairs closer together.
- Therefore, bond angles decrease.
Order of repulsion:
- Lone pair – lone pair > lone pair – bonding pair > bonding pair – bonding pair
2. Number of Electron Pairs
- Increasing number of electron pairs changes geometry and bond angles.
- More pairs → more repulsion → different arrangement.
3. Multiple Bonds
- Double and triple bonds contain more electron density.
- They repel more strongly than single bonds.
- This can slightly increase adjacent bond angles.
4. Electronegativity of Surrounding Atoms
- More electronegative atoms pull bonding electrons away from the central atom.
- This reduces repulsion between bonding pairs.
- Therefore, bond angle may decrease slightly.
Importance of Bond Angle
- Determines the shape of molecules.
- Affects molecular polarity and physical properties.
- Influences chemical reactivity.
Therefore, bond angle is essential for understanding molecular geometry and behaviour.
Example 1:
Explain why the bond angle in ammonia (107°) is smaller than in methane (109.5°).
▶️ Answer/Explanation
Methane has four bonding pairs and no lone pairs, giving a tetrahedral angle of 109.5°.
Ammonia has one lone pair and three bonding pairs.
Lone pairs repel more strongly than bonding pairs.
This pushes bonding pairs closer together.
Therefore, the bond angle decreases to about 107°.
Example 2:
Explain why the bond angle in water (104.5°) is smaller than in ammonia.
▶️ Answer/Explanation
Water has two lone pairs and two bonding pairs.
Ammonia has only one lone pair.
More lone pairs increase repulsion.
This pushes bonding pairs even closer together.
Therefore, the bond angle in water is smaller than in ammonia.
