Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.18 Shapes and bond angles -Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.18 Shapes and bond angles – Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.18 Shapes and bond angles – Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.18 Know and be able to explain the shapes of, and bond angles in, BeCl₂, BCl₃, CH₄, NH₃, NH₄⁺, H₂O, CO₂, gaseous PCl₅, SF₆ and C₂H₄.
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.18 Shapes and Bond Angles of Molecules and Ions
The shapes and bond angles of molecules and ions can be predicted using electron pair repulsion theory (VSEPR).
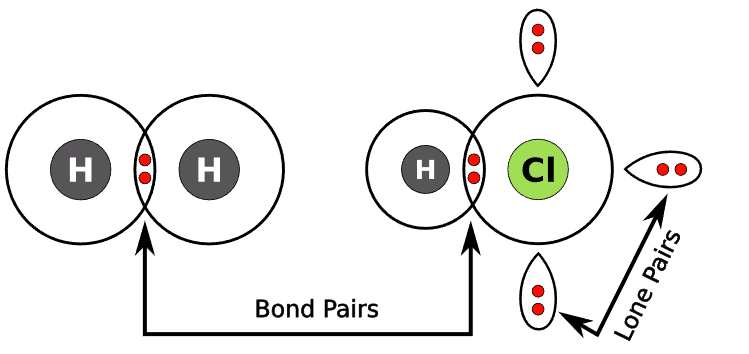
The arrangement depends on the number of bonding pairs and lone pairs around the central atom.
General Principles
- Electron pairs repel and arrange themselves as far apart as possible.
- Lone pairs repel more strongly than bonding pairs.
- Molecular shape is determined by the positions of atoms only.
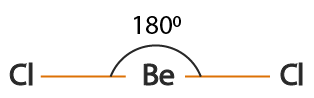
BeCl₂
2 bonding pairs, 0 lone pairs → linear
- Bond angle: 180°
- Electron pairs repel equally, giving maximum separation.
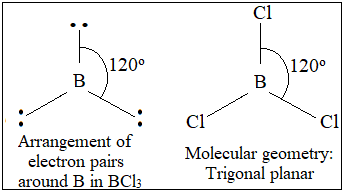
BCl₃
3 bonding pairs, 0 lone pairs → trigonal planar
- Bond angle: 120°
- Electron pairs spread evenly in one plane.
CH₄
4 bonding pairs, 0 lone pairs → tetrahedral
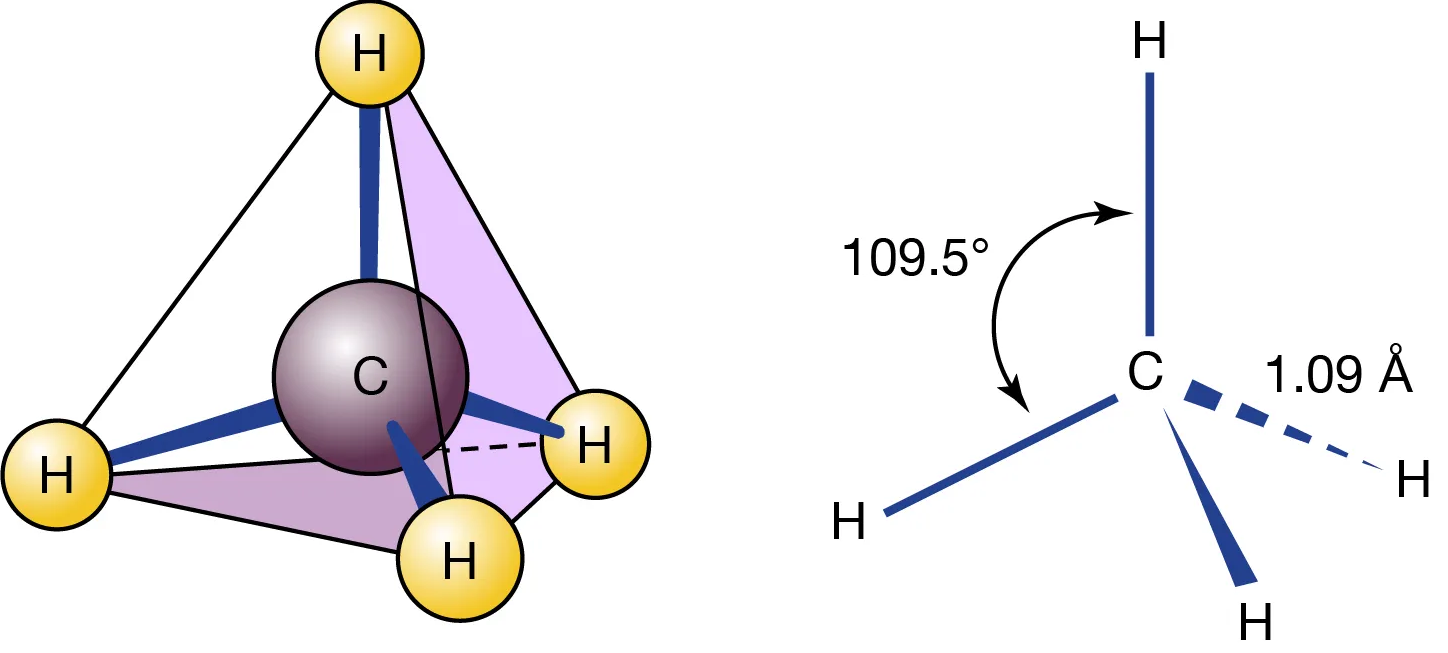
- Bond angle: 109.5°
- Symmetrical arrangement minimises repulsion.
NH₃
3 bonding pairs, 1 lone pair → trigonal pyramidal
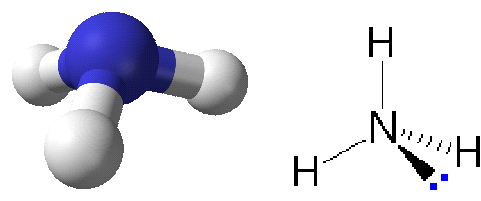
- Bond angle: ~107°
- Lone pair repels more strongly, reducing bond angle.
NH₄⁺
4 bonding pairs, 0 lone pairs → tetrahedral
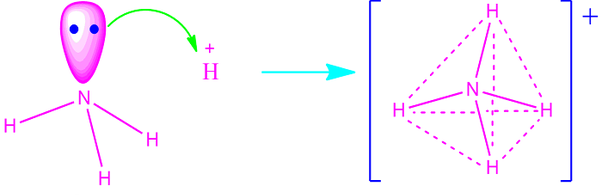
- Bond angle: 109.5°
- No lone pairs, so ideal tetrahedral angle.
H₂O
2 bonding pairs, 2 lone pairs → bent (V-shaped)
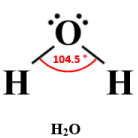
- Bond angle: ~104.5°
- Two lone pairs strongly repel bonding pairs, reducing angle further.
CO₂
2 bonding regions, 0 lone pairs → linear
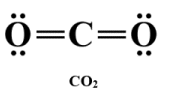
- Bond angle: 180°
- Double bonds count as one region of electron density.
PCl₅ (gaseous)
5 bonding pairs, 0 lone pairs → trigonal bipyramidal
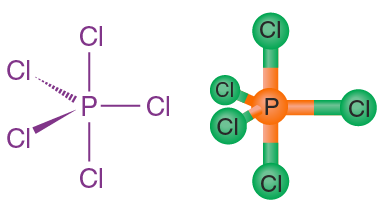
- Bond angles: 120° (equatorial), 90° (axial)
- Electron pairs arrange in two different planes.
SF₆
6 bonding pairs, 0 lone pairs → octahedral
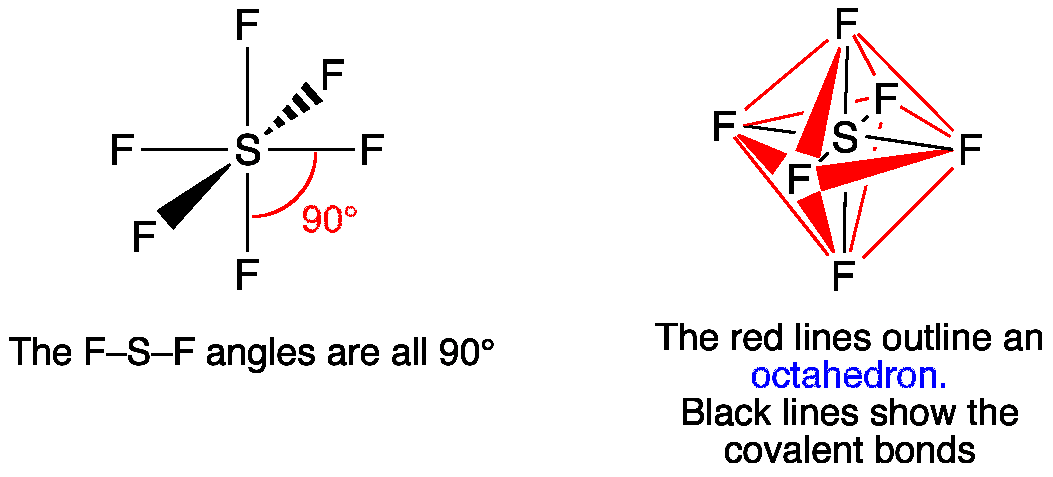
- Bond angle: 90°
- Symmetrical 3D arrangement.
C₂H₄ (Ethene)
Each carbon: 3 bonding regions, 0 lone pairs → trigonal planar
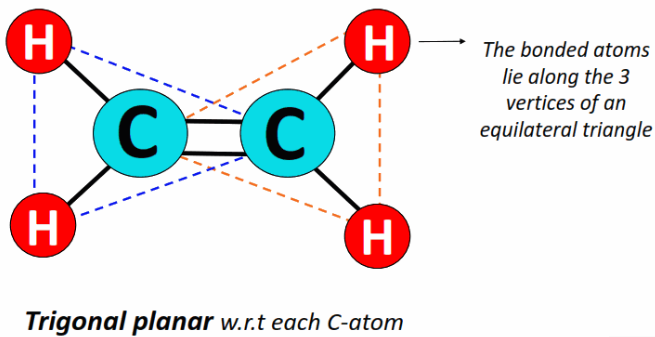
- Bond angle: 120°
- Double bond restricts rotation and keeps molecule planar.
Summary of Trends
- Increasing number of electron pairs changes geometry.
- Lone pairs reduce bond angles.
- Double bonds count as one region of electron density.
These shapes are essential for predicting molecular behaviour and properties.
Example 1:
Explain why ammonia has a smaller bond angle than methane.
▶️ Answer/Explanation
Methane has four bonding pairs and no lone pairs, giving a bond angle of 109.5°.
Ammonia has one lone pair and three bonding pairs.
Lone pairs repel more strongly than bonding pairs.
This reduces the bond angle to about 107°.
Example 2:
Explain why carbon dioxide is linear but water is bent.
▶️ Answer/Explanation
Carbon dioxide has two bonding regions and no lone pairs, so electron pairs arrange 180° apart.
Water has two bonding pairs and two lone pairs.
Lone pairs repel more strongly and distort the shape.
Therefore, water has a bent shape with a smaller bond angle.
