Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.19 Predicting analogous shapes-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.19 Predicting analogous shapes- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.19 Predicting analogous shapes- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.19 be able to apply the electron-pair repulsion theory to predict the shapes of, and bond angles in, molecules and ions analogous to those in 3.18
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.19 Applying Electron Pair Repulsion Theory
Electron-pair repulsion theory (VSEPR) can be applied to predict the shapes and bond angles of molecules and ions that are similar (analogous) to standard examples. The same principles used in 3.18 are extended to unfamiliar species.
Key Principle
The shape of a molecule depends on the number of electron pairs (bonding + lone pairs) around the central atom and how they arrange to minimise repulsion.
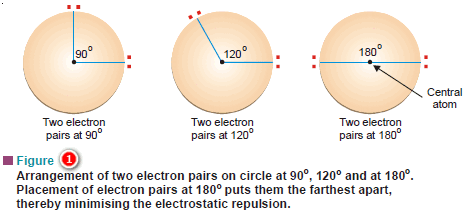
Step-by-Step Method
- Step 1: Draw the Lewis (dot-and-cross) structure.
- Step 2: Count the total number of electron pairs around the central atom.
- Step 3: Identify how many are bonding pairs and lone pairs.
- Step 4: Determine the electron pair geometry.
- Step 5: Adjust for lone pairs to find the actual molecular shape.
- Step 6: State the bond angle (and any deviations).
Important Rules
- Double and triple bonds count as one region of electron density.
- Lone pairs repel more strongly than bonding pairs.
- Molecular shape depends only on atom positions (not lone pairs).
- Bond angles decrease when lone pairs are present.
Applying to Analogous Molecules
Molecules with the same number of electron pairs have the same basic shape, even if the atoms are different.
- \( \mathrm{SiCl_4} \) is analogous to \( \mathrm{CH_4} \) → tetrahedral (109.5°).
- \( \mathrm{SO_2} \) is analogous to \( \mathrm{H_2O} \) → bent (less than 120°).
- \( \mathrm{NH_2^-} \) is analogous to \( \mathrm{H_2O} \) → bent (~104°).
- \( \mathrm{PF_5} \) is analogous to \( \mathrm{PCl_5} \) → trigonal bipyramidal.
- \( \mathrm{SF_4} \): 4 bonding pairs + 1 lone pair → seesaw shape.
Therefore, identifying similarities allows prediction of structure without memorising every molecule.
Effect of Lone Pairs in Analogous Structures
- Adding lone pairs changes shape but keeps the same electron geometry.
- For example:
- Tetrahedral → trigonal pyramidal → bent (as lone pairs increase).
- Bond angles decrease as lone pairs increase.
Summary
- Shape depends on number of electron pairs.
- Lone pairs reduce bond angles and alter shape.
- Similar electron pair arrangements lead to similar shapes.
- VSEPR can be applied to unfamiliar molecules using known patterns.
This allows prediction of molecular geometry without memorising every structure individually.
Example 1:
Predict the shape and bond angle of \( \mathrm{SiCl_4} \).
▶️ Answer/Explanation
Silicon has four bonding pairs and no lone pairs.
Electron pairs arrange tetrahedrally to minimise repulsion.
Therefore, the shape is tetrahedral.
The bond angle is approximately 109.5°.
Example 2:
Predict and explain the shape of \( \mathrm{SO_2} \).
▶️ Answer/Explanation
Sulfur has three regions of electron density (two bonding regions and one lone pair).
These arrange in a trigonal planar electron geometry.
The lone pair repels more strongly than bonding pairs.
This distorts the shape to bent.
The bond angle is slightly less than 120°.
