Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.20–3.21 Giant metallic lattice and electrostatic attraction-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.20–3.21 Giant metallic lattice and electrostatic attraction- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.20–3.21 Giant metallic lattice and electrostatic attraction- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.20 understand that metals consist of giant lattices of metal ions in a sea of delocalised electrons
3.21 know that metallic bonding is the strong electrostatic attraction between metal ions and the delocalised electrons
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.20 Metallic Bonding
Metals have a unique type of bonding and structure that explains their characteristic physical properties such as conductivity, malleability and high melting points.
Structure of Metals
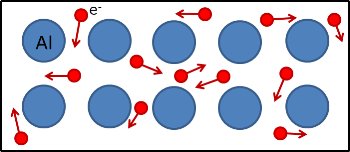
Metals consist of a giant lattice of positive metal ions surrounded by a sea of delocalised electrons.
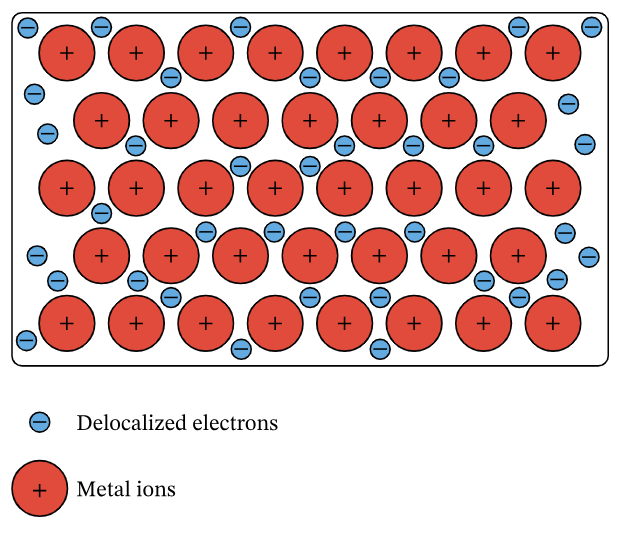
In this structure, outer shell electrons from metal atoms become delocalised, meaning they are no longer associated with a specific atom and are free to move throughout the entire lattice.
Metallic Bonding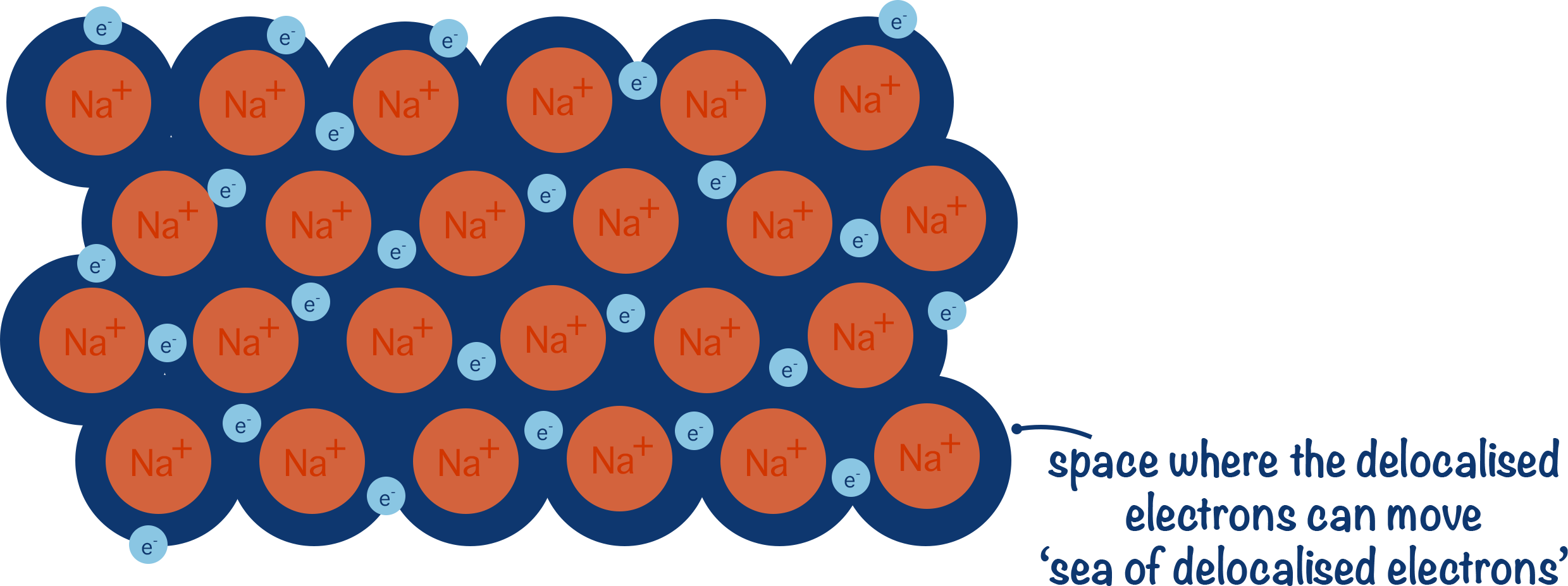
Metallic bonding is the strong electrostatic attraction between positive metal ions and delocalised electrons.
This attraction acts in all directions throughout the lattice, holding the structure together.
Key Features of Metallic Structure
- Positive metal ions are arranged in a regular, closely packed lattice.
- Delocalised electrons move freely throughout the structure.
- The bonding is non-directional and extends across the entire lattice.
Properties Explained by Metallic Bonding
Electrical conductivity
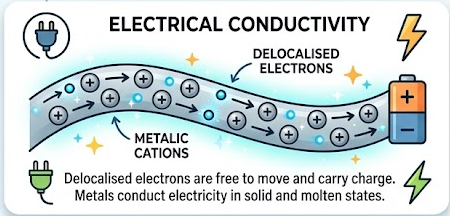
- Delocalised electrons are free to move and carry charge.
- Therefore, metals conduct electricity in both solid and molten states.
Thermal conductivity
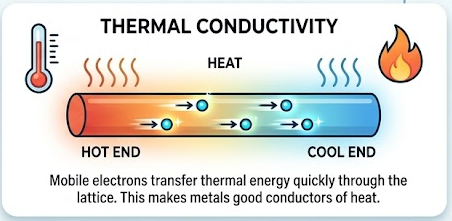
- Mobile electrons transfer thermal energy quickly through the lattice.
- This makes metals good conductors of heat.
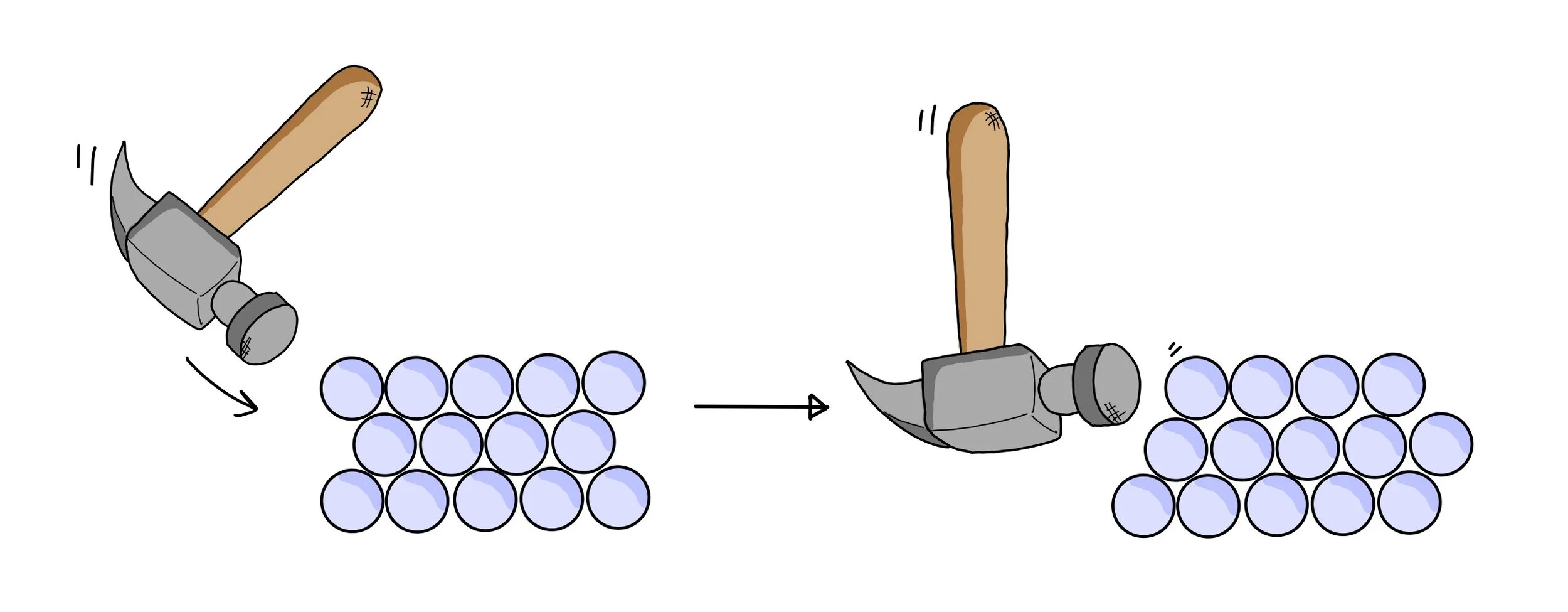
Malleability and ductility
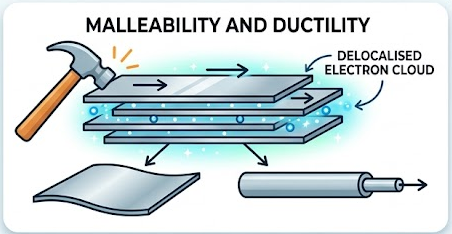
- Layers of metal ions can slide over each other without breaking the structure.
- The delocalised electrons maintain attraction between ions.
- Therefore, metals can be hammered into shape (malleable) and drawn into wires (ductile).
High melting and boiling points
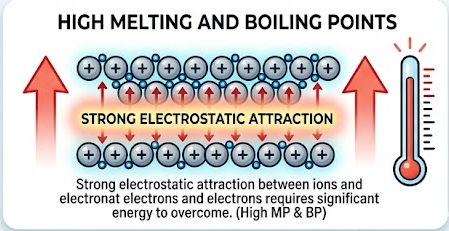
- Strong electrostatic attraction between ions and electrons requires significant energy to overcome.
- Therefore, most metals have high melting and boiling points.
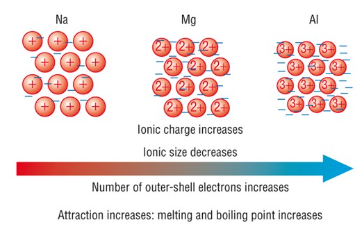
Factors Affecting Strength of Metallic Bonding
- Higher charge on metal ions increases attraction to electrons.
- More delocalised electrons strengthen the bonding.
- Smaller ionic radius increases charge density and strengthens attraction.
Therefore, metallic bonding explains the key physical properties of metals through the interaction between ions and delocalised electrons.
Example 1:
Explain why metals conduct electricity in the solid state.
▶️ Answer/Explanation
Metals contain delocalised electrons that are free to move.
These electrons can carry charge through the lattice.
Therefore, metals conduct electricity in the solid state.
Example 2:
Explain why magnesium has a higher melting point than sodium.
▶️ Answer/Explanation
Magnesium forms \( \mathrm{Mg^{2+}} \) ions, while sodium forms \( \mathrm{Na^+} \) ions.
Magnesium has more delocalised electrons and a higher charge.
This leads to stronger electrostatic attraction between ions and electrons.
Therefore, more energy is required to break the bonds.
Hence, magnesium has a higher melting point.
3.21 Metallic Bonding
Metallic bonding explains how metal atoms are held together in a solid metal and accounts for the characteristic properties of metals such as conductivity and strength.
Metallic Bonding
Metallic bonding is the strong electrostatic attraction between positive metal ions and delocalised electrons.
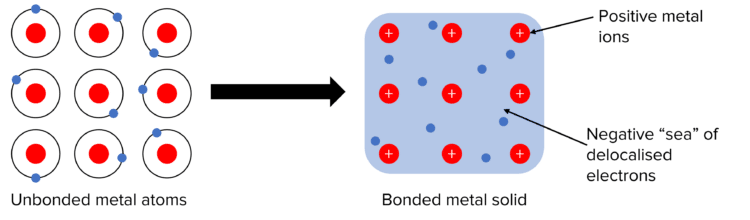
In a metal, atoms lose their outer shell electrons, forming a lattice of positive ions. The released electrons become delocalised and are free to move throughout the structure.
These delocalised electrons are attracted to all the positive ions in the lattice, creating a strong electrostatic force that holds the metal together.
Key Features
- Metal ions are arranged in a regular, closely packed lattice.
- Delocalised electrons move freely throughout the structure.
- The attraction is strong and acts in all directions.
Why the Bond is Strong
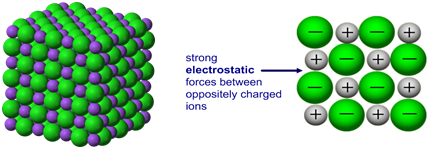
- There are many electrostatic attractions between ions and electrons.
- The attraction acts across the entire lattice, not just between specific particles.
- More delocalised electrons increase the strength of bonding.
Therefore, metallic bonding is a strong force that maintains the structure of metals.
Link to Properties
- Free electrons allow metals to conduct electricity.
- Strong attraction gives metals high melting points.
- Non-directional bonding allows layers to slide, making metals malleable.
Thus, metallic bonding explains both the structure and properties of metals.
Example 1:
State and explain what is meant by metallic bonding.
▶️ Answer/Explanation
Metallic bonding is the electrostatic attraction between positive metal ions and delocalised electrons.
Metal atoms lose electrons, forming a lattice of positive ions.
The electrons are free to move and are attracted to the ions.
This attraction holds the metal together.
Example 2:
Explain why metals have high melting points in terms of metallic bonding.
▶️ Answer/Explanation
There are strong electrostatic attractions between metal ions and delocalised electrons.
A large amount of energy is required to overcome these forces.
Therefore, metals have high melting points.