Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.22 Properties of metals-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.22 Properties of metals- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.22 Properties of metals- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.22 be able to use the models in 3.20 and 3.21 to interpret simple properties of metals, including electrical conductivity and high melting temperature
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.22 Properties of Metals Explained by Metallic Bonding
The models of metallic bonding (giant lattice of positive ions in a sea of delocalised electrons) can be used to explain key physical properties of metals, especially electrical conductivity and high melting temperature.
Electrical Conductivity
Metals conduct electricity because they contain delocalised electrons that are free to move throughout the lattice.
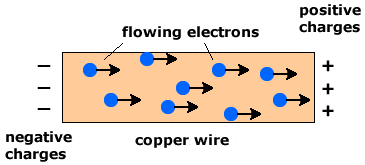
In the metallic structure, electrons are not bound to specific atoms and can move under the influence of an electric field.
- Delocalised electrons act as mobile charge carriers.
- When a potential difference is applied, electrons move towards the positive terminal.
- This flow of electrons constitutes an electric current.
- Therefore, metals conduct electricity in both solid and molten states.
The presence of freely moving electrons is the key reason for electrical conductivity in metals.
High Melting Temperature
Metals have high melting temperatures due to the strong electrostatic attraction between metal ions and delocalised electrons.
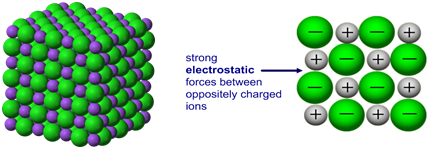
These forces act throughout the entire giant lattice and require a large amount of energy to overcome.
- Positive metal ions are strongly attracted to the surrounding sea of electrons.
- The attraction is strong and non-directional, acting in all directions.
- A large amount of energy is needed to separate the ions and electrons.
- Therefore, metals have high melting and boiling points.
Factors Affecting These Properties
- Metals with more delocalised electrons (e.g. Mg vs Na) conduct better and have stronger bonding.
- Higher charge on metal ions increases electrostatic attraction.
- Smaller ionic radius increases charge density and strengthens bonding.
Therefore, stronger metallic bonding leads to higher melting temperatures and often better conductivity.
Summary
- Electrical conductivity is due to mobile delocalised electrons.
- High melting temperature is due to strong electrostatic attraction in the lattice.
- Both properties are explained by the metallic bonding model.
Thus, the metallic bonding model provides a clear explanation for key physical properties of metals.
Example 1:
Explain why metals conduct electricity in both solid and molten states.
▶️ Answer/Explanation
Metals contain delocalised electrons that are free to move.
These electrons are not fixed in position in either solid or molten states.
When a potential difference is applied, they carry charge through the metal.
Therefore, metals conduct electricity in both states.
Example 2:
Explain why aluminium has a higher melting point than sodium.
▶️ Answer/Explanation
Aluminium forms \( \mathrm{Al^{3+}} \) ions, while sodium forms \( \mathrm{Na^+} \) ions.
Aluminium has more delocalised electrons and a higher charge.
This results in stronger electrostatic attraction between ions and electrons.
More energy is required to overcome these forces.
Therefore, aluminium has a higher melting point.
