Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.3 Dot-and-cross diagrams (ions)-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.3 Dot-and-cross diagrams (ions)- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.3 Dot-and-cross diagrams (ions)- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.3 be able to draw dot-and-cross diagrams to show electrons in cations and anions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.3 Dot-and-Cross Diagrams for Ions
Dot-and-cross diagrams are used to represent the arrangement of electrons in atoms and ions. They show how electrons are lost or gained during ion formation and help visualise the resulting charges on ions.
In these diagrams:
- Dots (•) and crosses (×) are used to distinguish electrons from different atoms.
- Square brackets are used around ions.
- The charge of the ion is shown outside the brackets.
Dot-and-Cross Diagrams for Cations
Cations are formed when atoms lose electrons.
When drawing diagrams for cations:
- Show the original outer-shell electrons being removed.
- Draw the resulting ion with a full outer shell (if applicable).
- Place the ion in square brackets and include the positive charge.
Example: Sodium forming a sodium ion
Na → Na\( ^+ \) + e\( ^- \)
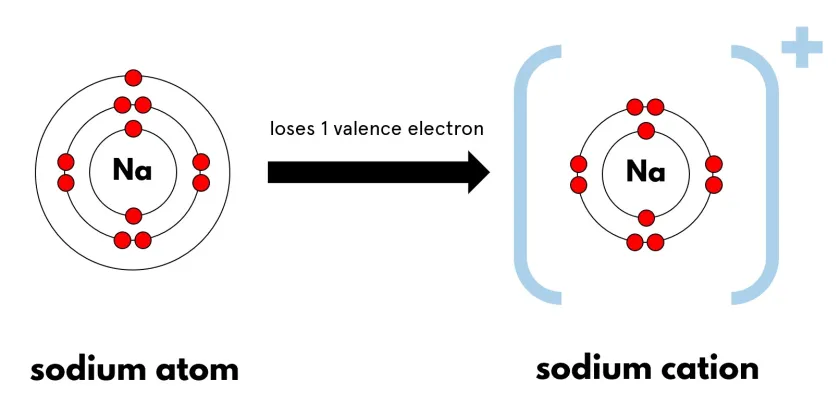
Final ion: [Na]\( ^+ \) (outer shell now full)
Example: Magnesium forming a magnesium ion
Mg → Mg\( ^{2+} \) + 2e\( ^- \)
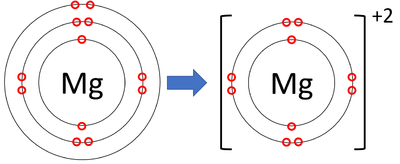
Final ion: [Mg]\( ^{2+} \)
Dot-and-Cross Diagrams for Anions
Anions are formed when atoms gain electrons.
When drawing diagrams for anions:
- Show the additional electrons gained (usually from another atom).
- Use different symbols (dots/crosses) to indicate electrons from different sources.
- Draw a full outer shell (usually 8 electrons).
- Enclose the ion in square brackets with the negative charge.
Example: Chlorine forming a chloride ion
Cl + e\( ^- \) → Cl\( ^- \)
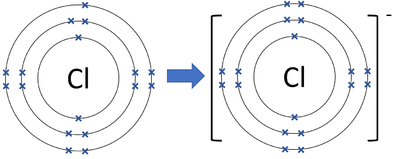
Final ion: [Cl]\( ^- \) with 8 outer electrons
Example: Oxygen forming an oxide ion
O + 2e\( ^- \) → O\( ^{2-} \)
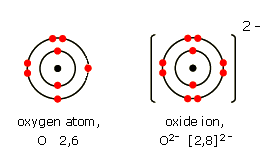
Final ion: [O]\( ^{2-} \) with 8 outer electrons
Key Features of Correct Diagrams
- Use square brackets around ions.
- Show charges clearly outside the brackets.
- Ensure the correct number of electrons in the outer shell (usually 8).
- Distinguish electrons from different atoms using dots and crosses.
These diagrams help show how atoms achieve stable electronic configurations through ion formation.
Common Dot-and-Cross Diagrams for Important Ions
The following table summarises important ions and their dot-and-cross representations. These are commonly required in exams and help illustrate how atoms gain or lose electrons to achieve a full outer shell.
| Ion | Electron Change | Dot-and-Cross Diagram (Outer Shell) | Explanation |
|---|---|---|---|
| \( \mathrm{Na^+} \) | Loses 1 electron | 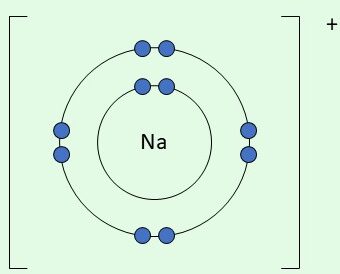 | Outer shell becomes full after losing 1 electron |
| \( \mathrm{Mg^{2+}} \) | Loses 2 electrons | 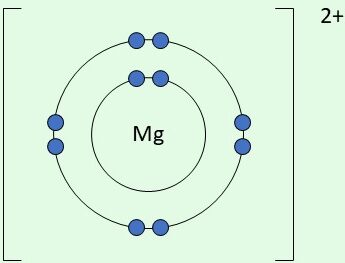 | Achieves stable noble gas configuration |
| \( \mathrm{Al^{3+}} \) | Loses 3 electrons | 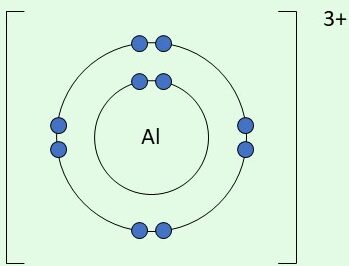 | Empty outer shell after losing 3 electrons |
| \( \mathrm{Cl^-} \) | Gains 1 electron | 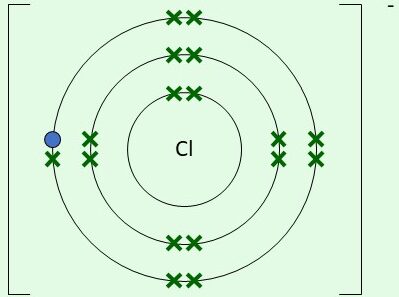 | 7 original electrons (dots) + 1 gained (cross) |
| \( \mathrm{O^{2-}} \) | Gains 2 electrons | 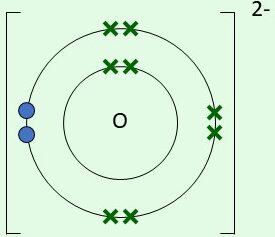 | 6 original electrons + 2 gained electrons |
| \( \mathrm{P^{3-}} \) | Gains 3 electrons | 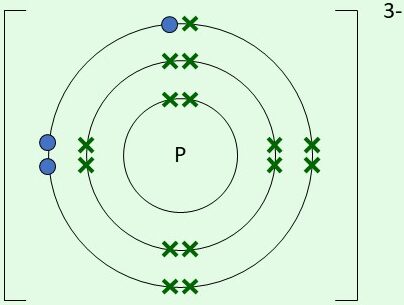 | 5 original electrons + 3 gained electrons |
Key Observations
- Metals form positive ions by losing electrons (no outer-shell electrons shown after loss).
- Non-metals form negative ions by gaining electrons to complete an octet.
- Dots represent original electrons, crosses represent electrons gained from another atom.
- All ions shown achieve a stable noble gas configuration.
Example 1:
Draw a dot-and-cross diagram to show the formation of \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) ions.
▶️ Answer/Explanation
Sodium loses one electron to form \( \mathrm{Na^+} \), and chlorine gains one electron to form \( \mathrm{Cl^-} \).
[Na]\( ^+ \)
[Cl]\( ^- \) with 8 outer electrons (7 original + 1 gained)
The transferred electron is shown as a cross if sodium’s electrons are represented by crosses.
Example 2:
Draw a dot-and-cross diagram for the oxide ion \( \mathrm{O^{2-}} \), showing the origin of electrons.
▶️ Answer/Explanation
Oxygen gains two electrons to complete its outer shell.
[O]\( ^{2-} \) with 6 original electrons (dots) and 2 gained electrons (crosses)
The full outer shell contains 8 electrons.
