Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.4 Ionic lattices-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.4 Ionic lattices- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.4 Ionic lattices- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.4 be able to describe ionic crystals as giant lattices of ions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.4 Ionic Crystals as Giant Lattices
Ionic crystals are made up of a giant lattice of ions arranged in a regular, repeating three-dimensional structure. This structure extends throughout the entire crystal and is not made up of individual molecules.
The lattice consists of positive ions (cations) and negative ions (anions) held together by strong electrostatic forces of attraction.
Structure of an Ionic Lattice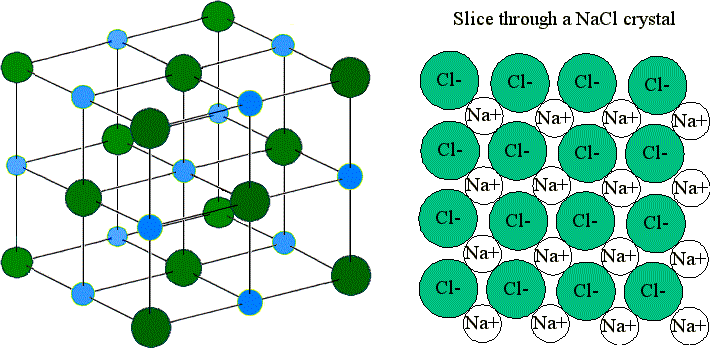
- Each ion is surrounded by ions of opposite charge.
- The arrangement is highly ordered and repeats in all directions.
- There are strong electrostatic forces between oppositely charged ions acting in all directions.
For example, in sodium chloride:
Each \( \mathrm{Na^+} \) ion is surrounded by six \( \mathrm{Cl^-} \) ions.
Each \( \mathrm{Cl^-} \) ion is surrounded by six \( \mathrm{Na^+} \) ions.
This arrangement is known as a giant ionic lattice.
Nature of Ionic Bonding in the Lattice
Ionic bonding is the strong electrostatic attraction between oppositely charged ions formed after the transfer of electrons from one atom to another.
This attraction is non-directional, meaning it acts equally in all directions throughout the lattice rather than between specific pairs of ions.
As a result:
- Each ion is attracted to many oppositely charged ions surrounding it, not just one.
- The strength of the attraction depends on the charge of the ions and the distance between them.
For example:
- Ions with higher charges (e.g. \( \mathrm{Mg^{2+}} \), \( \mathrm{O^{2-}} \)) form stronger ionic bonds than ions with lower charges.
- Smaller ions allow ions to be closer together, increasing electrostatic attraction.
Because these forces act throughout the entire structure, the ionic lattice is extremely strong and stable.
Importantly, there are no discrete molecules in ionic compounds—only a continuous three-dimensional network of ions.
Properties Explained by the Giant Lattice
High melting and boiling points
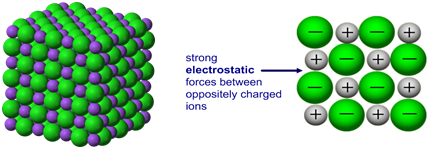
- The strong electrostatic forces of attraction between oppositely charged ions require a large amount of energy to overcome.
- These forces act in all directions across the entire lattice, not just between individual ions.
- As a result, ionic compounds have very high melting and boiling points.
The greater the ionic charge and the smaller the ionic radius, the stronger the attraction and the higher the melting point.
Brittleness
Ionic crystals are brittle due to the arrangement of ions in the lattice.
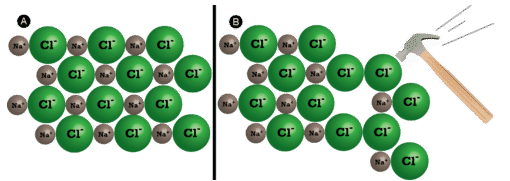
When a force is applied:
- Layers of ions may shift relative to each other.
- This can bring ions of the same charge (e.g. \( \mathrm{Na^+} \) next to \( \mathrm{Na^+} \)) into alignment.
- Strong electrostatic repulsion between like charges occurs.
- This causes the lattice to break suddenly along planes, leading to the crystal shattering.
Therefore, ionic compounds do not bend—they fracture.
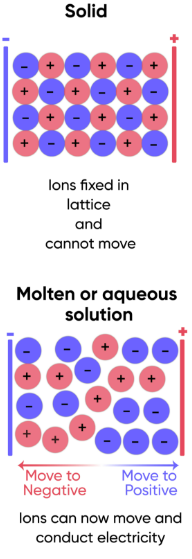
Electrical conductivity
- In the solid state, ions are fixed in position within the lattice and cannot move.
- Since charge carriers must be mobile for electrical conduction, solid ionic compounds do not conduct electricity.
However, when the compound is molten or dissolved in water:
- The lattice breaks down and ions become free to move.
- These mobile ions can carry charge through the liquid or solution.
- Therefore, molten and aqueous ionic compounds conduct electricity.
Solubility
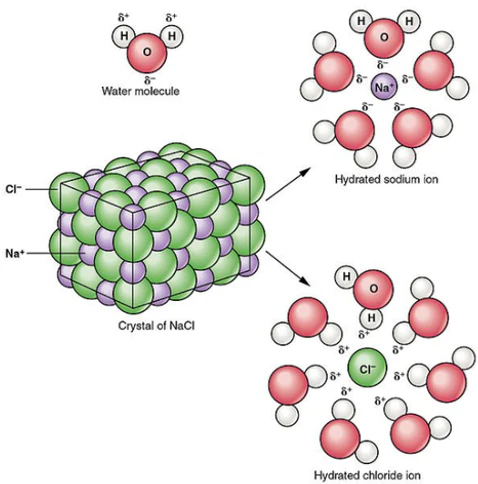
- Many ionic compounds are soluble in water because polar water molecules interact with ions.
- Water molecules surround ions (hydration), reducing the electrostatic forces between them.
- This allows ions to separate and dissolve into solution.
However, not all ionic compounds are soluble—this depends on the balance between lattice energy and hydration energy.
Example 1:
Explain why sodium chloride has a high melting point.
▶️ Answer/Explanation
Sodium chloride has a giant ionic lattice with strong electrostatic forces between \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) ions.
A large amount of energy is required to overcome these forces.
Therefore, it has a high melting point.
Example 2:
Explain why ionic crystals are brittle.
▶️ Answer/Explanation
When a force is applied, layers of ions may shift.
This can cause ions of the same charge to come into contact.
The strong repulsion between like charges causes the lattice to break.
Therefore, ionic crystals are brittle.
