Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.5 Electrostatic attraction in ionic bonding-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.5 Electrostatic attraction in ionic bonding- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.5 Electrostatic attraction in ionic bonding- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.5 know that ionic bonding is the result of strong net electrostatic attraction between ions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.5 Ionic Bonding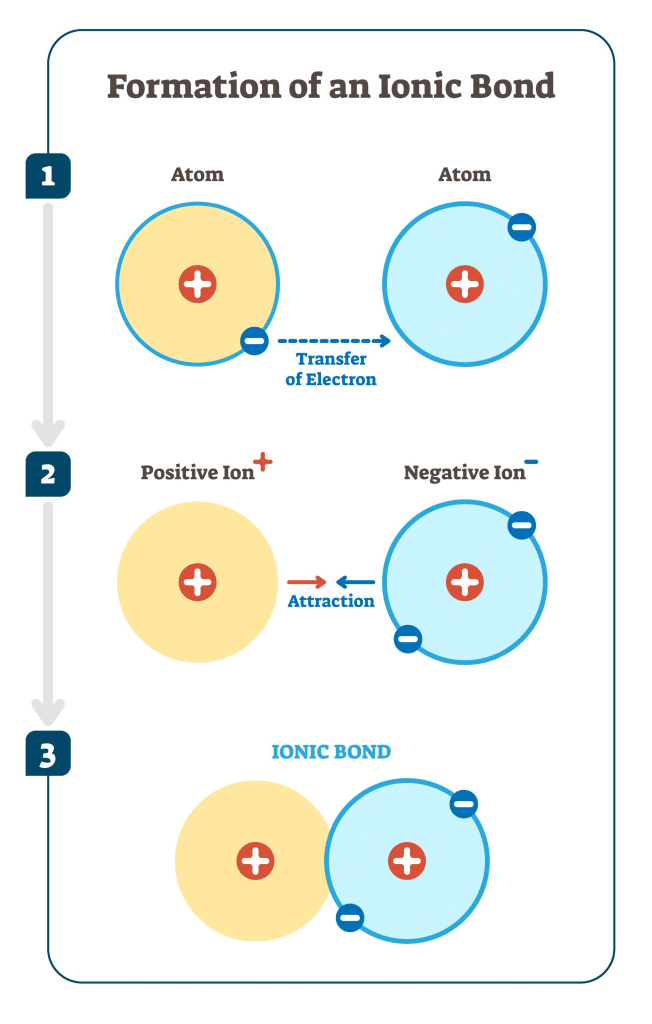
Ionic bonding explains how atoms combine to form ionic compounds through the formation of charged particles and the forces between them.
Ionic Bonding
Ionic bonding is the strong net electrostatic attraction between oppositely charged ions.
This attraction arises after electrons are transferred from one atom to another, forming ions with opposite charges.
Formation of Ions
- Metals lose electrons to form positive ions (cations).
- Non-metals gain electrons to form negative ions (anions).
This transfer of electrons allows atoms to achieve a stable electron configuration, usually a full outer shell similar to a noble gas.
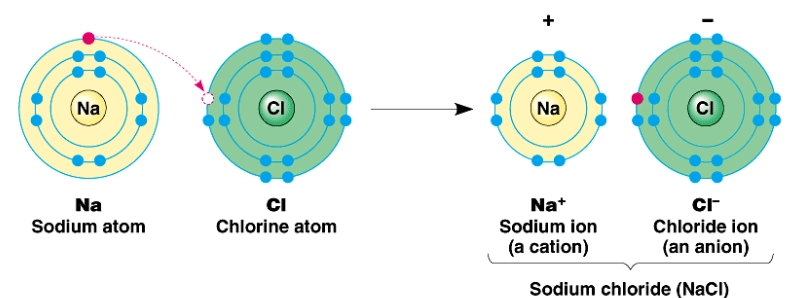
Example: Formation of Sodium Chloride
\( \mathrm{Na \rightarrow Na^+ + e^-} \)
\( \mathrm{Cl + e^- \rightarrow Cl^-} \)
The oppositely charged ions formed are then held together by strong electrostatic attraction.
\( \mathrm{Na^+ \ \cdots \ Cl^-} \)
This attraction acts in all directions and leads to the formation of a giant ionic lattice rather than individual molecules.
Nature of the Electrostatic Attraction
- The attraction is strong because it occurs between oppositely charged ions.
- The force is non-directional, meaning each ion is attracted to many surrounding ions.
- Each ion experiences both attractive and repulsive forces, but the overall effect is a strong net attraction.
- The strength of this attraction depends on ionic charge and the distance between ions.
Greater ionic charge and smaller ionic radius result in stronger electrostatic attraction and therefore stronger ionic bonding.
Example 1:
Explain what is meant by ionic bonding.
▶️ Answer/Explanation
Ionic bonding is the strong electrostatic attraction between oppositely charged ions.
These ions are formed by the transfer of electrons from one atom to another.
The attraction acts in all directions within a lattice structure.
Example 2:
Explain why magnesium oxide has stronger ionic bonding than sodium chloride.
▶️ Answer/Explanation
Magnesium oxide contains \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \) ions, while sodium chloride contains \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) ions.
The ions in magnesium oxide have higher charges.
This leads to stronger electrostatic attraction between ions.
Therefore, magnesium oxide has stronger ionic bonding.
