Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.6 Ionic radius and charge effects-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.6 Ionic radius and charge effects- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.6 Ionic radius and charge effects- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.6 understand the effects of ionic radius and ionic charge on the strength of ionic bonding
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.6 Effects of Ionic Radius and Ionic Charge on Ionic Bond Strength
The strength of ionic bonding depends on the magnitude of the electrostatic forces of attraction between oppositely charged ions in a giant ionic lattice. These forces act in all directions throughout the lattice and determine properties such as melting point and lattice stability.
The strength of this attraction is mainly affected by two key factors: the charge of the ions and the distance between the ions (ionic radius).
Effect of Ionic Charge
Ionic bonding becomes stronger as the magnitude of the ionic charge increases.
This is because electrostatic attraction depends directly on the size of the charges on the ions. Ions with larger charges exert stronger attractive forces on oppositely charged ions.
- Ions with higher positive or negative charges produce stronger electrostatic forces of attraction.
- For example, \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \) have double the charge compared to \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \).
- This leads to a much stronger attraction between ions in magnesium oxide than in sodium chloride.
- As a result, more energy is required to separate the ions in the lattice.
- This gives compounds with highly charged ions higher melting and boiling points and greater lattice stability.
Therefore, increasing ionic charge significantly increases the strength of ionic bonding.
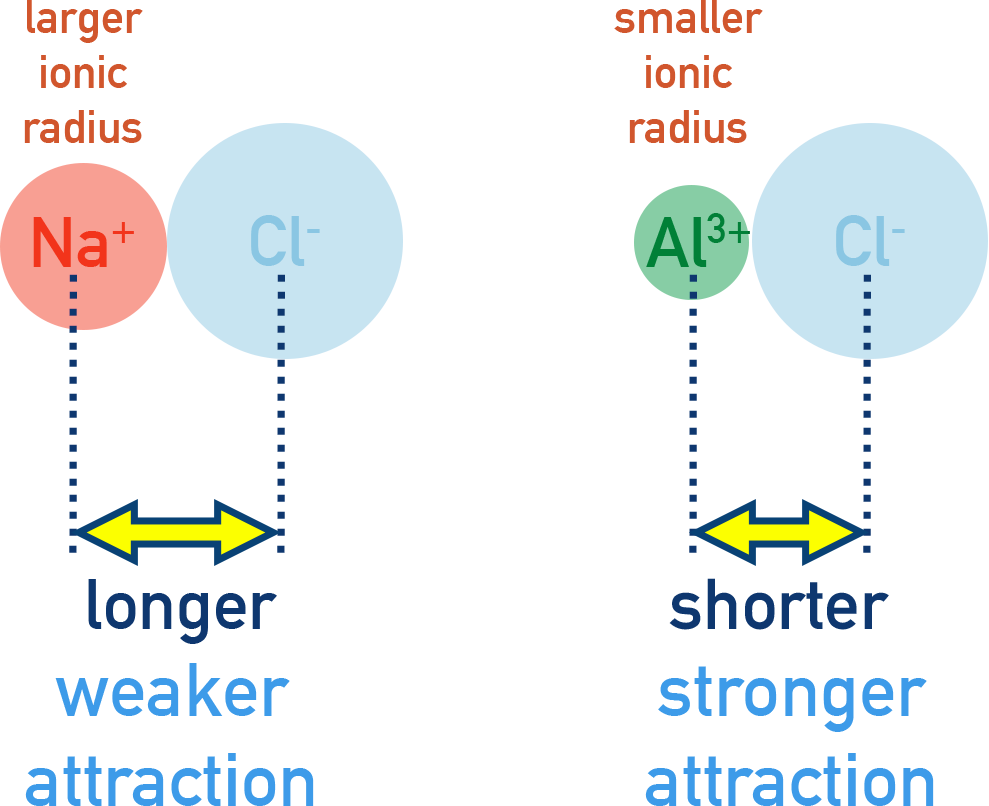
Effect of Ionic Radius
Ionic bonding becomes stronger as the ionic radius decreases.
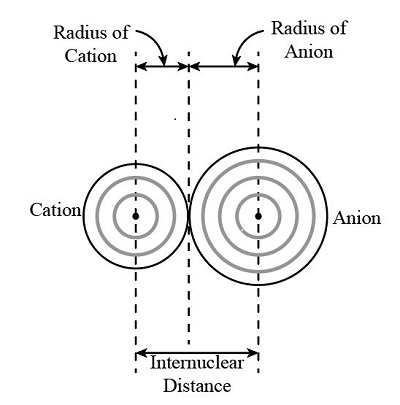
The ionic radius determines the distance between the nuclei of oppositely charged ions. The smaller the ions, the closer they can approach each other.
Since electrostatic attraction increases as distance decreases, smaller ions result in stronger attraction.
- Smaller ions allow charges to be closer together, increasing the force of attraction.
- Larger ions increase the distance between charges, weakening the attraction.
- For example, \( \mathrm{Li^+} \) is smaller than \( \mathrm{K^+} \), so lithium fluoride has stronger ionic bonding than potassium fluoride.
- Down a group, ionic radius increases, so ionic bond strength generally decreases.
- Across a period, ions tend to be smaller, which can increase ionic bond strength.
Therefore, decreasing ionic radius increases the strength of ionic bonding.
Combined Effect on Ionic Bond Strength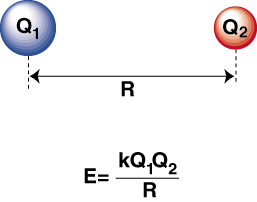
- Ionic bond strength depends on both ionic charge and ionic radius together.
- The strongest ionic bonds occur when ions have high charges and small radii.
- These conditions maximise electrostatic attraction between ions.
- This results in very strong ionic lattices with high lattice energies.
- Consequently, such compounds have very high melting points and are very stable.
In summary, ionic bonding is strongest when the electrostatic attraction is maximised by high ionic charge and small ionic radius.
Example 1:
Explain why magnesium oxide has a much higher melting point than sodium chloride.
▶️ Answer/Explanation
Magnesium oxide contains \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \) ions, whereas sodium chloride contains \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) ions.
The ions in magnesium oxide have higher charges.
Higher charges result in stronger electrostatic attraction between oppositely charged ions.
Therefore, much more energy is required to overcome these forces.
This leads to a significantly higher melting point for magnesium oxide.
Example 2:
Explain the trend in melting point from lithium fluoride to potassium fluoride.
▶️ Answer/Explanation
Lithium fluoride and potassium fluoride contain ions with the same charges.
However, the ionic radius of the metal cation increases from \( \mathrm{Li^+} \) to \( \mathrm{K^+} \).
This increases the distance between oppositely charged ions.
As the distance increases, electrostatic attraction becomes weaker.
Therefore, lattice strength decreases down the group.
As a result, melting point decreases from lithium fluoride to potassium fluoride.
