Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.7 Isoelectronic ions and trends-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.7 Isoelectronic ions and trends- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.7 Isoelectronic ions and trends- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.7 understand reasons for the trends in ionic radii down a group in the Periodic Table, and for a set of isoelectronic ions, including N3− to Al3+
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.7 Trends in Ionic Radii
The size of ions (ionic radius) is an important factor that affects the strength of ionic bonding and the properties of ionic compounds. Ionic radius depends on the number of electron shells and the attraction between the nucleus and the electrons.
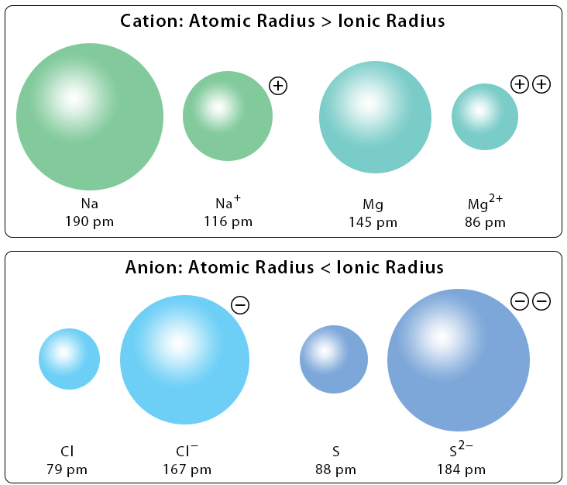
Trends in ionic radii can be explained by considering changes down a group and across a series of isoelectronic ions.
Trend in Ionic Radius Down a Group
Ionic radius increases down a group in the Periodic Table.
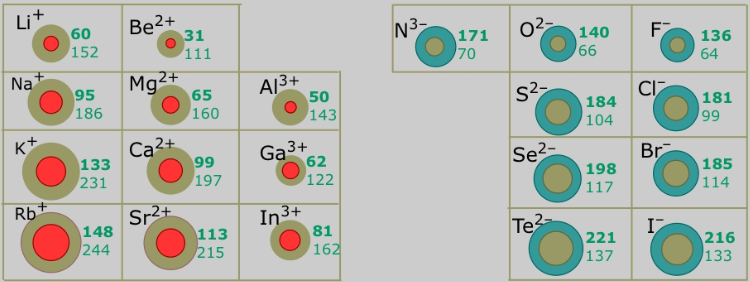
This trend occurs because additional electron shells are added as you move down the group.
- Each step down a group adds a new principal energy level (electron shell).
- This increases the distance between the nucleus and the outermost electrons.
- Increased shielding effect reduces the attraction between the nucleus and outer electrons.
- As a result, the outer electrons are held less strongly and the ion becomes larger.
- Therefore, ionic radius increases down the group.
For example, the ionic radius increases from \( \mathrm{Li^+} \) to \( \mathrm{Na^+} \) to \( \mathrm{K^+} \).
Isoelectronic Ions
Isoelectronic ions are ions that have the same number of electrons but different nuclear charges.
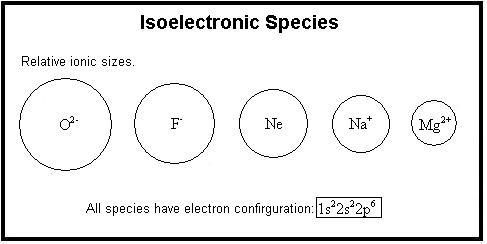
Since these ions have identical electron configurations, differences in their radii are due to the number of protons in the nucleus.
Trend in Isoelectronic Ions (N³⁻ to Al³⁺)
Ionic radius decreases as nuclear charge increases across an isoelectronic series.
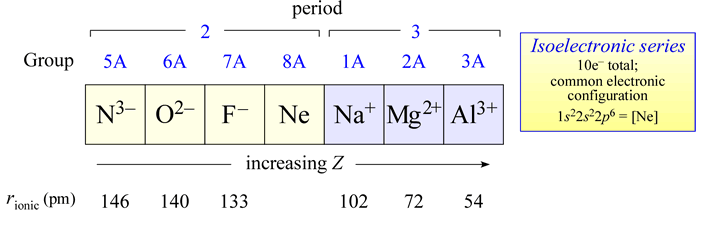
In the series \( \mathrm{N^{3-}, O^{2-}, F^-, Na^+, Mg^{2+}, Al^{3+}} \), all ions have the same number of electrons but different numbers of protons.
- As the number of protons increases, the nuclear charge increases.
- The stronger positive charge pulls electrons closer to the nucleus.
- This results in a decrease in ionic radius across the series.
- Negative ions (anions) are larger because of increased electron-electron repulsion.
- Positive ions (cations) are smaller because electrons are pulled more strongly by the nucleus.
Therefore, ionic radius decreases from \( \mathrm{N^{3-}} \) to \( \mathrm{Al^{3+}} \).
Summary of Key Ideas
- Down a group: ionic radius increases due to more electron shells and increased shielding.
- Across an isoelectronic series: ionic radius decreases due to increasing nuclear charge.
- Larger ions have weaker electrostatic attraction; smaller ions have stronger attraction.
These trends help explain variations in ionic bond strength and properties of ionic compounds.
Example 1:
Explain why the ionic radius of potassium ions is larger than that of sodium ions.
▶️ Answer/Explanation
Potassium ions have an additional electron shell compared to sodium ions.
This increases the distance between the nucleus and the outer electrons.
Increased shielding reduces the attraction between the nucleus and electrons.
Therefore, potassium ions have a larger ionic radius than sodium ions.
Example 2:
Explain why \( \mathrm{Al^{3+}} \) has a much smaller ionic radius than \( \mathrm{N^{3-}} \).
▶️ Answer/Explanation
Both ions are isoelectronic and have the same number of electrons.
However, \( \mathrm{Al^{3+}} \) has more protons than \( \mathrm{N^{3-}} \).
The greater nuclear charge in \( \mathrm{Al^{3+}} \) pulls the electrons closer to the nucleus.
In contrast, \( \mathrm{N^{3-}} \) has fewer protons and greater electron-electron repulsion.
Therefore, \( \mathrm{Al^{3+}} \) is much smaller than \( \mathrm{N^{3-}} \).
