Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 3.8–3.9 Polarisation, polarising power and polarisability-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.8–3.9 Polarisation, polarising power and polarisability- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 3.8–3.9 Polarisation, polarising power and polarisability- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
3.8 understand the meaning of the term ‘polarisation’ as applied to ions
3.9 understand that the polarising power of a cation depends on its radius and charge, and the polarisability of an anion also depends on its radius and charge
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
3.8 Polarisation of Ions
Polarisation helps explain how ionic compounds can show some covalent character instead of being purely ionic. It describes how the electron distribution in an ion can be distorted by the presence of another ion.
Polarisation
Polarisation is the distortion of the electron cloud of an anion by the electric field of a nearby cation.
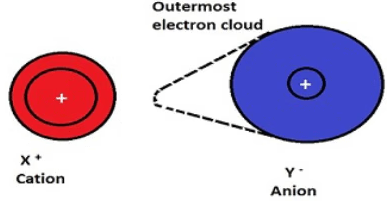
In a purely ionic bond, electron density is completely transferred and remains centred on the anion. However, in reality, the positive ion can attract and distort this electron cloud.
This distortion means the electron density is no longer evenly distributed, and some electron density is pulled towards the cation.
How Polarisation Occurs
- A cation has a positive charge and attracts electrons.
- An anion has a cloud of electrons that can be distorted.
- The cation pulls electron density towards itself, distorting the anion’s electron cloud.
- This results in uneven electron distribution between the ions.
Factors Affecting Polarisation
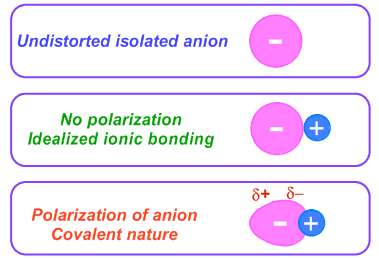
The extent of polarisation depends on the polarising power of the cation and the polarisability of the anion.
Polarising Power of Cation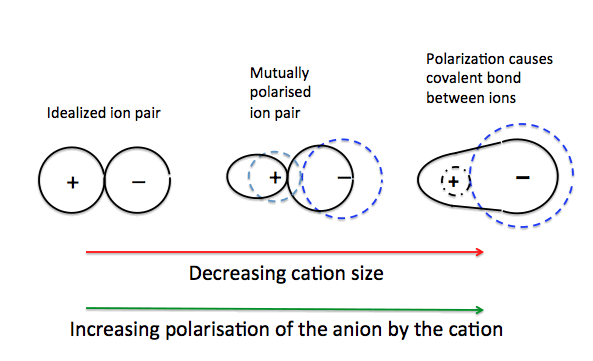
- Small cations have high charge density and can strongly attract electrons.
- Highly charged cations (e.g. \( \mathrm{Al^{3+}} \)) have greater polarising power.
- Therefore, small and highly charged cations cause greater distortion.
Polarisability of Anion
- Large anions have more diffuse electron clouds that are easier to distort.
- Highly charged anions (e.g. \( \mathrm{I^-} \)) are more easily polarised.
- Therefore, large anions are more polarisable.
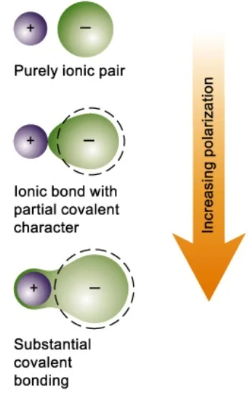
Effect of Polarisation on Bonding
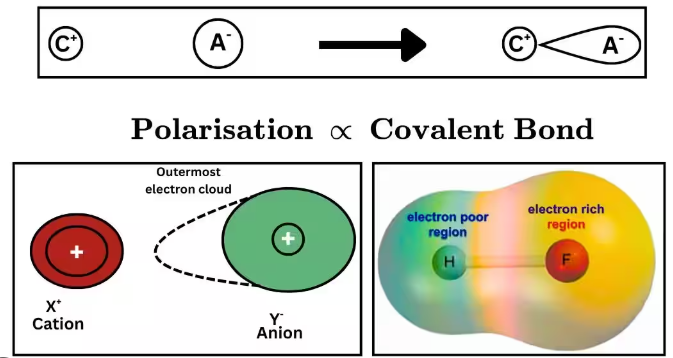
- Greater polarisation leads to increased covalent character in the bond.
- The bond becomes less purely ionic as electron density is shared more.
- Highly polarised compounds may show properties closer to covalent substances.
Therefore, polarisation explains why some ionic compounds are not completely ionic and instead have partial covalent character.
Example 1:
Explain what is meant by polarisation in ionic bonding.
▶️ Answer/Explanation
Polarisation is the distortion of the electron cloud of an anion by a nearby cation.
The positive cation attracts electron density towards itself.
This results in uneven distribution of electrons.
This distortion introduces covalent character into the ionic bond.
Example 2:
Explain why aluminium chloride shows more covalent character than sodium chloride.
▶️ Answer/Explanation
Aluminium ions \( \mathrm{Al^{3+}} \) are small and highly charged.
This gives them high polarising power.
They strongly distort the electron cloud of chloride ions.
Sodium ions \( \mathrm{Na^+} \) have lower charge and larger size, so they have lower polarising power.
Therefore, aluminium chloride has greater covalent character than sodium chloride.
3.9 Polarising Power and Polarisability
The extent of polarisation in an ionic compound depends on how strongly a cation can distort an anion and how easily the anion’s electron cloud can be distorted. These are described as the polarising power of the cation and the polarisability of the anion.
Both depend on the charge and radius of the ions, which together determine charge density and how strongly electrons are attracted or distorted.
Polarising Power of a Cation
Polarising power is the ability of a cation to distort the electron cloud of an anion.
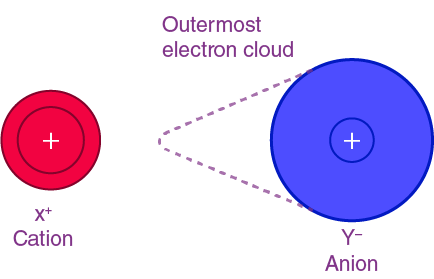
This depends on how strongly the cation attracts electrons, which is determined by its charge density (charge per unit volume).
- Cations with higher charge have stronger electrostatic attraction for electrons.
- Smaller cations have electrons closer to the nucleus, resulting in higher charge density.
- Therefore, small, highly charged cations have the greatest polarising power.
- For example, \( \mathrm{Al^{3+}} \) has much greater polarising power than \( \mathrm{Na^+} \).
As polarising power increases, distortion of the anion increases and the bond gains more covalent character.
Polarisability of an Anion
Polarisability is the ease with which the electron cloud of an anion can be distorted.
This depends on how loosely the electrons are held by the nucleus and how spread out the electron cloud is.
- Larger anions have more diffuse electron clouds, which are easier to distort.
- Anions with higher negative charge have greater electron-electron repulsion.
- This reduces the attraction between the nucleus and electrons, making distortion easier.
- Therefore, large, highly charged anions are the most polarisable.
- For example, \( \mathrm{I^-} \) is more polarisable than \( \mathrm{F^-} \).
Greater polarisability leads to greater distortion and increased covalent character in the bond.
Combined Effect
- Maximum polarisation occurs when a small, highly charged cation interacts with a large, highly charged anion.
- This results in significant distortion of electron density.
- The bond becomes less ionic and more covalent in character.
- This concept is summarised by Fajans’ rules.
Therefore, polarising power and polarisability together determine the extent of polarisation and the nature of bonding in ionic compounds.
Example 1:
Explain why aluminium oxide has more covalent character than magnesium oxide.
▶️ Answer/Explanation
Aluminium ions \( \mathrm{Al^{3+}} \) have a higher charge and smaller radius than \( \mathrm{Mg^{2+}} \).
This gives aluminium ions greater polarising power.
They distort the electron cloud of oxide ions more strongly.
This increases covalent character in aluminium oxide.
Therefore, aluminium oxide is more covalent than magnesium oxide.
Example 2:
Explain why silver iodide is more covalent than silver fluoride.
▶️ Answer/Explanation
Both compounds contain the same cation \( \mathrm{Ag^+} \), so polarising power is similar.
However, iodide ions \( \mathrm{I^-} \) are much larger than fluoride ions \( \mathrm{F^-} \).
Larger anions are more polarisable and their electron clouds are more easily distorted.
Therefore, silver iodide experiences greater polarisation.
This results in greater covalent character compared to silver fluoride.
Further Suggested Practical: Migration of Ions
This practical demonstrates the movement of ions in an electric field, providing evidence that ions in solution are mobile charge carriers. It supports the concept of electrical conductivity in aqueous ionic compounds.
Principle
When an electric current is passed through an electrolyte, cations move towards the negative electrode (cathode) and anions move towards the positive electrode (anode).
This movement of ions is known as migration of ions and can be observed using coloured ions in solution.
Method 1: U-tube with Copper(II) Chromate Solution
- A U-tube is filled with a solution of copper(II) chromate.
- Inert electrodes (e.g. graphite or platinum) are placed in each arm of the U-tube.
- The electrodes are connected to a direct current power supply.
- When the current is switched on, coloured ions begin to move.
Observations
- Blue \( \mathrm{Cu^{2+}} \) ions move towards the cathode (negative electrode).
- Yellow \( \mathrm{CrO_4^{2-}} \) ions move towards the anode (positive electrode).
- The movement can be seen as coloured regions shifting in opposite directions.
Method 2: Microscope Slide with Potassium Manganate(VII)
- A drop of potassium manganate(VII) solution is placed on a microscope slide.
- Two electrodes are positioned on either side of the drop.
- A small current is passed through the solution.
Observations
- Purple \( \mathrm{MnO_4^-} \) ions move towards the anode (positive electrode).
- The movement of colour clearly shows ion migration.
Explanation
- Ions in solution are free to move and carry charge.
- Positive ions are attracted to the negative electrode, while negative ions are attracted to the positive electrode.
- This confirms that electrical conduction in solutions occurs via movement of ions.
Conclusion
The experiment demonstrates that ions in aqueous solution migrate towards oppositely charged electrodes, confirming that ionic compounds conduct electricity when dissolved due to the presence of mobile ions.
Example 1:
Explain why copper(II) ions move towards the cathode in this experiment.
▶️ Answer/Explanation
Copper(II) ions are positively charged (\( \mathrm{Cu^{2+}} \)).
Opposite charges attract, so they are attracted to the negatively charged cathode.
Therefore, they migrate towards the cathode.
Example 2:
Explain why solid ionic compounds do not conduct electricity but aqueous solutions do.
▶️ Answer/Explanation
In solid ionic compounds, ions are fixed in a lattice and cannot move.
Since there are no mobile charge carriers, electricity cannot flow.
In aqueous solutions, the lattice breaks down and ions become free to move.
These mobile ions carry charge through the solution.
Therefore, ionic solutions conduct electricity.
