Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.1–4.3 Hazard, risk assessment and risk reduction-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.1–4.3 Hazard, risk assessment and risk reduction- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.1–4.3 Hazard, risk assessment and risk reduction- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.1 understand the difference between hazard and risk
4.2 understand the hazards associated with organic compounds and why it is necessary to carry out risk assessments when dealing with potentially hazardous materials
4.3 be able to suggest ways in which risks can be reduced and reactions carried out safely, for example:
i working on a smaller scale
ii taking precautions specific to the hazard
iii using an alternative method that involves less hazardous substances
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.1 Hazard and Risk
In chemistry, it is important to distinguish between hazard and risk when handling substances. These terms are related but have different meanings.
Hazard
A hazard is a substance or situation with the potential to cause harm.

Hazards describe the inherent properties of a substance that make it dangerous, regardless of how it is used.
- For example, a chemical may be toxic, flammable or corrosive.
- These properties exist even if the substance is not currently causing harm.
- Therefore, a hazard refers to the potential danger.
Risk
Risk is the likelihood that a hazard will actually cause harm under specific conditions.
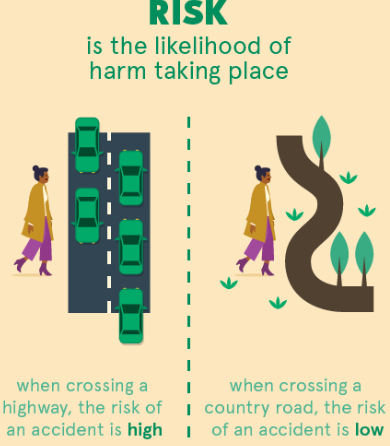
Risk depends on how the substance is used, including factors such as quantity, concentration, and exposure.
- A highly hazardous substance may present low risk if handled safely.
- A less hazardous substance may present higher risk if used carelessly.
- Therefore, risk involves both hazard and exposure.
Key Differences
- Hazard = potential to cause harm.
- Risk = likelihood of harm occurring.
- Hazard is an inherent property; risk depends on conditions.
Therefore, a substance can be hazardous but pose little risk if proper precautions are taken.
Example 1:
Explain the difference between hazard and risk using a toxic chemical as an example.
▶️ Answer/Explanation
A toxic chemical is hazardous because it can cause harm to health.
However, the risk depends on exposure.
If handled in small amounts with protective equipment, the risk is low.
Therefore, hazard is the potential for harm, while risk is the likelihood of harm occurring.
Example 2:
Explain why a flammable substance may have low risk in a laboratory.
▶️ Answer/Explanation
A flammable substance is hazardous because it can catch fire easily.
However, if it is kept away from flames and used in small amounts, the chance of ignition is low.
Therefore, the risk is low even though the hazard remains.
4.2 Hazards of Organic Compounds and Risk Assessment
Many organic compounds can pose significant hazards due to their chemical properties. Understanding these hazards and carrying out risk assessments is essential to ensure safe handling in the laboratory.
Hazards Associated with Organic Compounds
Organic compounds often contain covalent bonds and can have properties that make them dangerous under certain conditions.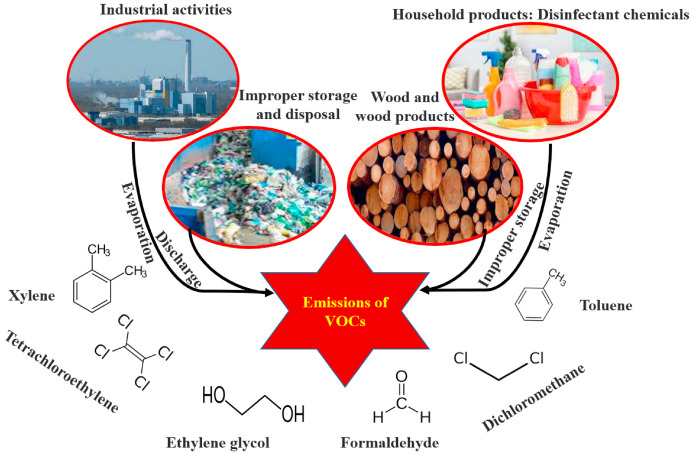
Flammability
- Many organic compounds (e.g. hydrocarbons, alcohols) are highly flammable.
- They can ignite easily when exposed to heat, sparks or flames.
- Vapours may also be flammable and form explosive mixtures with air.
Toxicity
- Some organic compounds are toxic and can harm the body if inhaled, ingested or absorbed through the skin.
- Long-term exposure may lead to serious health effects.
Volatility
- Many organic liquids are volatile and evaporate easily.
- This increases the risk of inhalation and fire hazards.
Corrosiveness / Irritation
- Some organic substances can irritate or damage skin, eyes or respiratory system.
Environmental Hazards
- Certain organic compounds are harmful to aquatic life and may persist in the environment.
Therefore, organic compounds can present multiple hazards depending on their structure and properties.
Risk Assessment
A risk assessment is the process of identifying hazards, evaluating risks, and implementing control measures to reduce the likelihood of harm.
Steps in Risk Assessment
- Identify the hazards (e.g. flammable, toxic substances).
- Assess the risk (likelihood and severity of harm).
- Decide on control measures (e.g. safety equipment, procedures).
- Review and monitor safety during the experiment.
Why Risk Assessments are Necessary
- To minimise the chance of accidents such as fires, poisoning or injury.
- To ensure safe handling, storage and disposal of chemicals.
- To protect both individuals and the environment.
- To comply with laboratory safety regulations.
Therefore, risk assessment is essential when working with potentially hazardous organic compounds.
Examples of Control Measures
- Use of fume cupboards to avoid inhalation of vapours.
- Wearing protective equipment such as gloves and goggles.
- Keeping flammable substances away from ignition sources.
- Using small quantities to reduce risk.
These measures reduce risk even when hazards cannot be eliminated.
Example 1:
Explain why many organic compounds are considered fire hazards.
▶️ Answer/Explanation
Many organic compounds are volatile and evaporate easily.
Their vapours can mix with air to form flammable mixtures.
They also contain carbon and hydrogen, which burn readily.
Therefore, they can ignite easily and pose fire hazards.
Example 2:
Explain why a risk assessment is important when working with a toxic organic solvent.
▶️ Answer/Explanation
A toxic solvent can cause harm if inhaled or absorbed through the skin.
A risk assessment identifies these hazards and evaluates the level of risk.
Appropriate control measures such as using a fume cupboard and wearing gloves can be implemented.
This reduces exposure and minimises the chance of harm.
Therefore, risk assessment ensures safe handling of hazardous substances.
4.3 Reducing Risk and Carrying Out Reactions Safely
When working with chemicals, especially organic compounds, it is essential to reduce risk by applying appropriate safety measures. These measures aim to minimise the likelihood of harm while still allowing the experiment to be carried out effectively.
Key Principle
Risk can be reduced by limiting exposure to hazards and controlling experimental conditions.
(i) Working on a Smaller Scale
Using smaller quantities of chemicals reduces the potential harm in case of an accident.
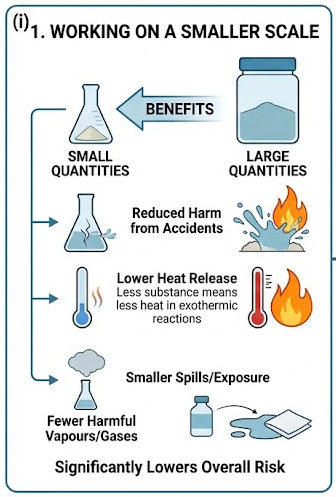
- Less substance means less heat released in exothermic reactions.
- Reduced production of harmful vapours or gases.
- Smaller spills or exposure lead to lower risk.
Therefore, working on a smaller scale significantly lowers the overall risk of the experiment.
(ii) Taking Precautions Specific to the Hazard
Safety measures should be chosen based on the specific hazard of the substance being used.
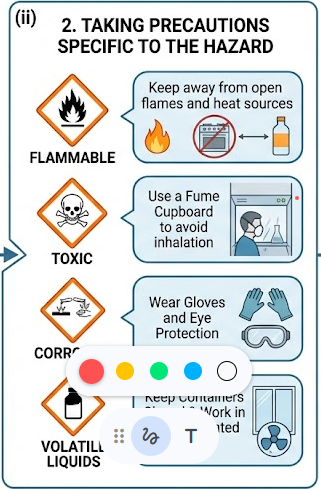
- Flammable substances: keep away from open flames and heat sources.
- Toxic substances: use a fume cupboard to avoid inhalation.
- Corrosive substances: wear gloves and eye protection.
- Volatile liquids: keep containers closed and work in well-ventilated areas.
These targeted precautions reduce the specific risks associated with each hazard.
(iii) Using Alternative Methods or Substances
Risk can be reduced by choosing safer alternatives where possible.
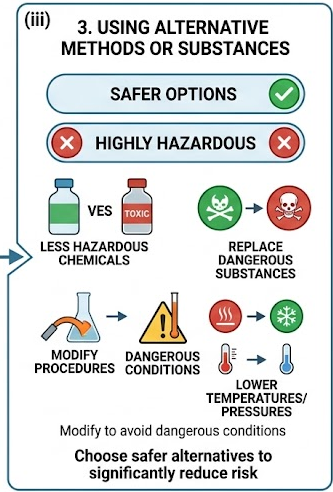
- Use less hazardous chemicals that perform the same function.
- Replace highly toxic or flammable substances with safer options.
- Modify procedures to avoid dangerous conditions (e.g. lower temperatures or pressures).
Therefore, selecting safer alternatives can significantly reduce risk without affecting the outcome.
Additional General Safety Measures
- Wear appropriate personal protective equipment (PPE), such as goggles and lab coats.
- Follow correct laboratory procedures and instructions.
- Ensure proper storage and disposal of chemicals.
- Be aware of emergency procedures in case of accidents.
Summary
- Risk can be reduced by minimising quantity, controlling hazards, and using safer alternatives.
- Safety measures must be appropriate to the specific hazard.
- Careful planning and precautions ensure safe experimental practice.
Thus, applying these strategies allows reactions to be carried out safely while minimising potential harm.
Example 1:
Explain how working on a smaller scale reduces risk in a chemical reaction.
▶️ Answer/Explanation
Using smaller quantities reduces the amount of energy released in reactions.
It also limits the amount of hazardous substances involved.
Therefore, any accident such as spills or reactions will have less severe consequences.
Example 2:
Suggest safety precautions when working with a flammable organic solvent.
▶️ Answer/Explanation
The solvent should be kept away from open flames and heat sources.
It should be used in a well-ventilated area to prevent vapour build-up.
Containers should be kept closed when not in use.
These measures reduce the risk of ignition and fire.
