Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.10 Structural isomerism-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.10 Structural isomerism- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.10 Structural isomerism- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.10 understand the term ‘structural isomerism’ and be able to draw the structural isomers of organic molecules, given their molecular formula
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.10 Structural Isomerism
In organic chemistry, compounds can have the same molecular formula but different structures. This phenomenon is known as structural isomerism.
Definition
Structural isomerism occurs when compounds have the same molecular formula but different structural formulae (different connectivity of atoms).
These differences in structure lead to differences in physical properties and sometimes chemical properties.
Types of Structural Isomerism
1. Chain Isomerism
- Different arrangements of the carbon skeleton.
- One may be straight-chain, another branched.
Example: C₄H₁₀
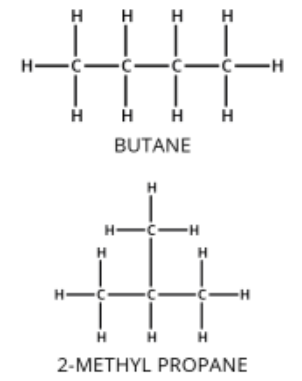
- Butane: CH₃CH₂CH₂CH₃
- 2-methylpropane: CH₃CH(CH₃)CH₃
2. Position Isomerism
- Same carbon skeleton, but functional group or multiple bond is in a different position.
Example: C₃H₇OH
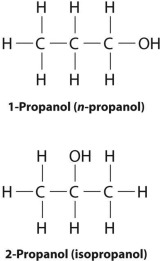
- Propan-1-ol: CH₃CH₂CH₂OH
- Propan-2-ol: CH₃CHOHCH₃
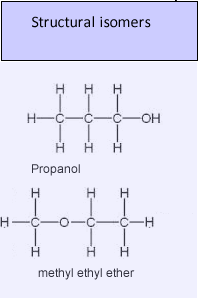
3. Functional Group Isomerism
- Same molecular formula but different functional groups.
Example: C₂H₆O
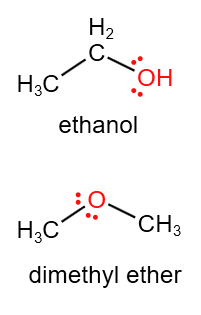
- Ethanol: CH₃CH₂OH (alcohol)
- Methoxymethane: CH₃OCH₃ (ether)
Drawing Structural Isomers
Step-by-Step Method
- Step 1: Identify the molecular formula.
- Step 2: Draw the longest possible carbon chain.
- Step 3: Rearrange the carbon skeleton (branching).
- Step 4: Change positions of functional groups if applicable.
- Step 5: Ensure all valencies are satisfied (C = 4 bonds).
- Step 6: Check that each structure is unique (no duplicates).
Important Points
- Each isomer must have the same molecular formula.
- Only the arrangement of atoms differs.
- Avoid drawing the same structure in a different orientation.
Summary
- Structural isomers have same molecular formula but different structures.
- Types include chain, position and functional group isomerism.
- Drawing isomers requires systematic rearrangement of atoms.
Therefore, structural isomerism explains how compounds with identical formulas can have different structures and properties.
Example 1:
Draw all structural isomers of C₄H₁₀.
▶️ Answer/Explanation
There are two isomers:
CH₃CH₂CH₂CH₃ (butane)
CH₃CH(CH₃)CH₃ (2-methylpropane)
Example 2:
Explain why structural isomers have different physical properties.
▶️ Answer/Explanation
Structural isomers have different arrangements of atoms.
This affects intermolecular forces between molecules.
Therefore, they have different boiling points and other physical properties.
