Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.12 Alkanes as fuels-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.12 Alkanes as fuels- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.12 Alkanes as fuels- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.12 know that alkanes are used as fuels and obtained from the fractional distillation, cracking and reforming of crude oil, and be able to write equations for these reactions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.12 Alkanes as Fuels and Their Production from Crude Oil
Alkanes are widely used as fuels and are obtained from crude oil through processes such as fractional distillation, cracking and reforming.
Alkanes as Fuels
Alkanes are good fuels because they burn in oxygen to release energy.
- Combustion produces heat energy.
- Used in domestic heating, transport and electricity generation.
Complete Combustion
In excess oxygen, alkanes burn completely to form carbon dioxide and water.
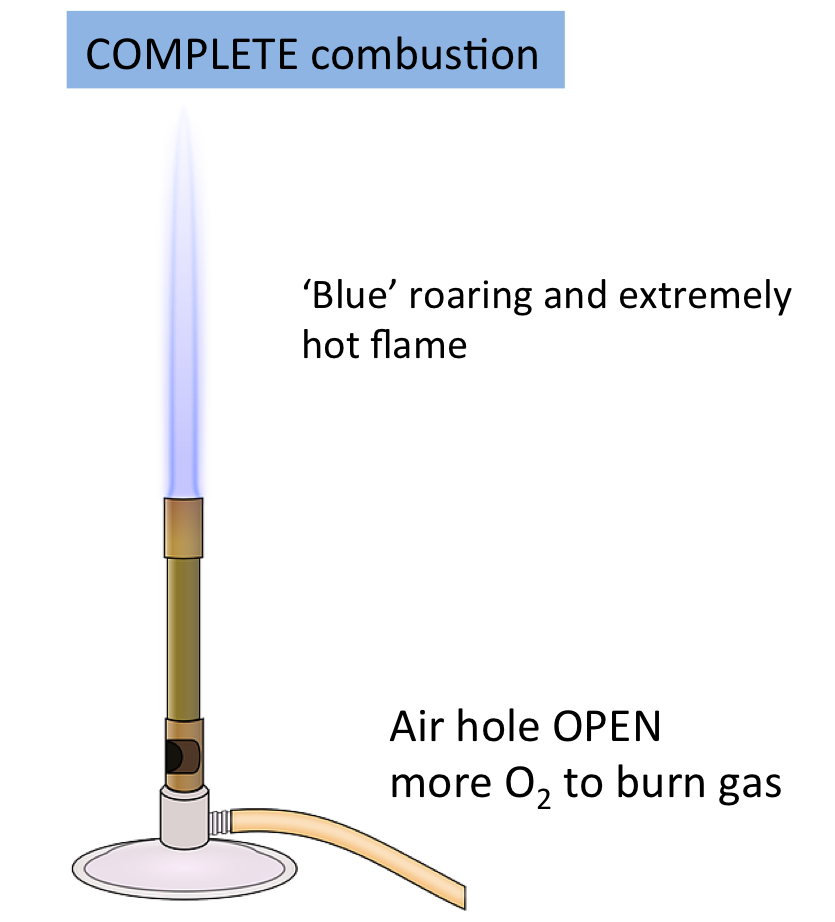
\( \mathrm{C_nH_{2n+2} + O_2 \rightarrow CO_2 + H_2O} \)
Example
- \( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Incomplete Combustion
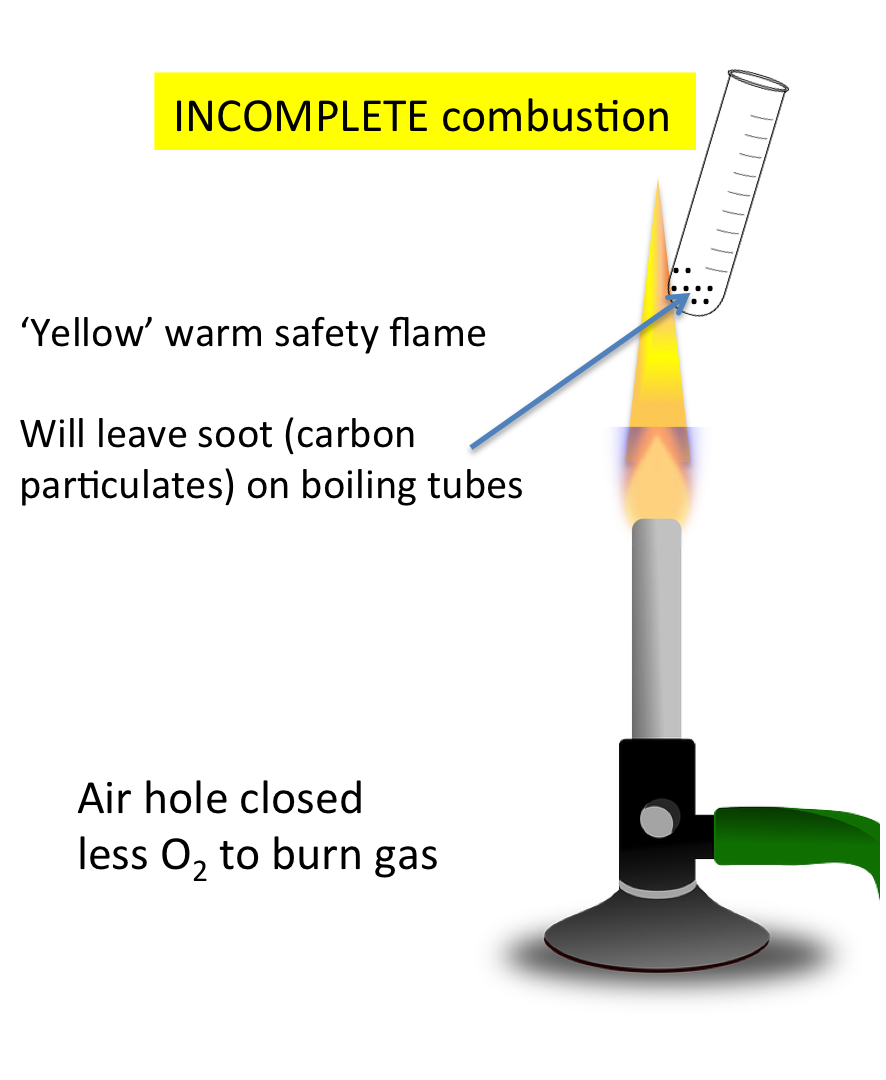
- Occurs when oxygen supply is limited.
- Produces carbon monoxide (CO) or carbon (soot).
Fractional Distillation of Crude Oil
Crude oil is a mixture of hydrocarbons that is separated into fractions based on boiling points.
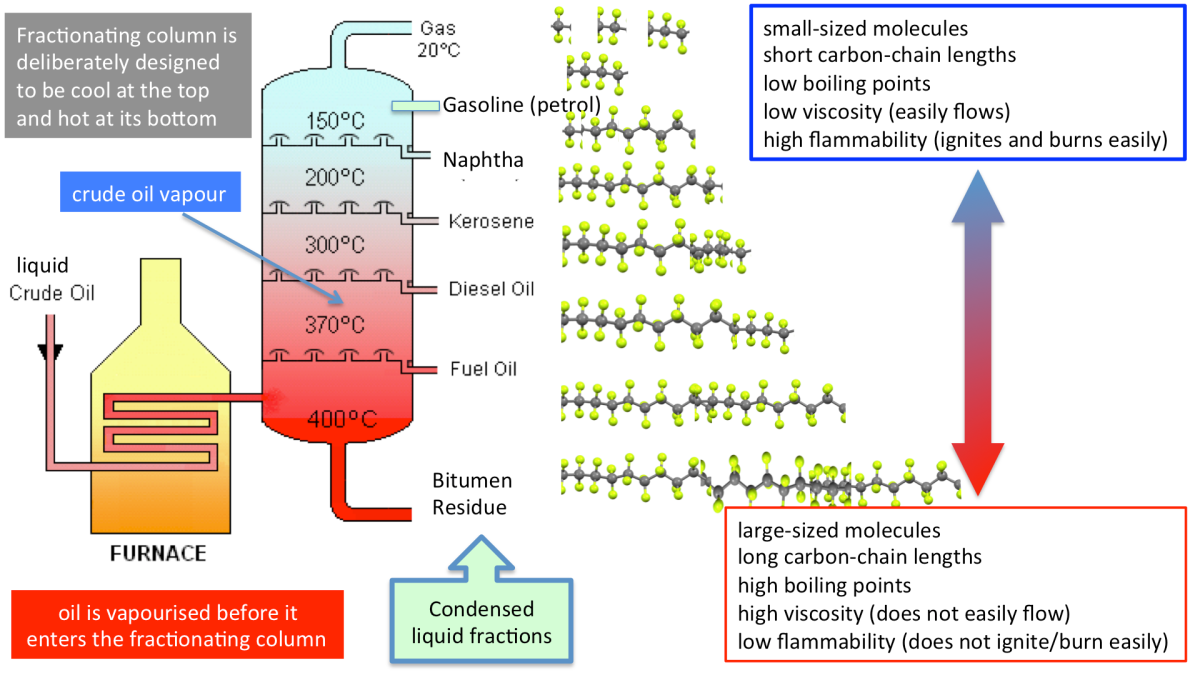
- Heated and vaporised.
- Vapours condense at different heights in the column.
- Produces fractions such as gases, petrol, kerosene and diesel.
This is a physical process (no chemical reaction).
Cracking
Cracking is the breaking of large hydrocarbon molecules into smaller alkanes and alkenes.
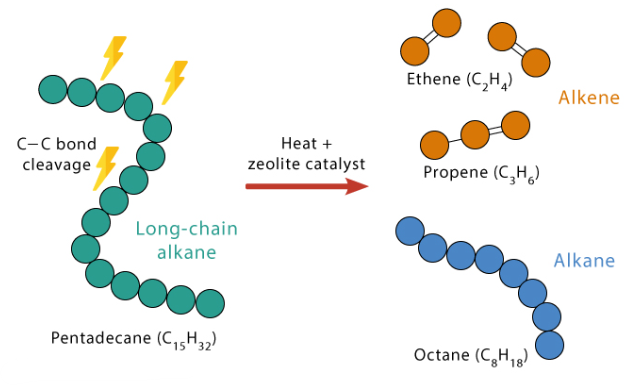
- Uses heat and/or catalysts.
- Produces more useful fuels and alkenes.
Example Equation
- \( \mathrm{C_{10}H_{22} \rightarrow C_8H_{18} + C_2H_4} \)
Therefore, cracking increases the supply of smaller, more useful hydrocarbons.
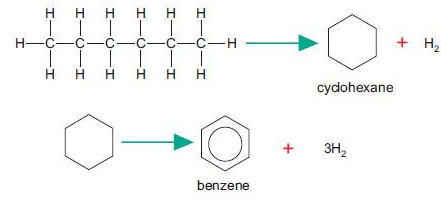
Reforming
Reforming is the rearrangement of hydrocarbon molecules to form branched or cyclic hydrocarbons.
- Improves fuel quality (higher octane number).
- Produces hydrogen gas as a by-product.
Example Equation
Therefore, reforming converts straight-chain alkanes into more efficient fuels.
Summary
- Alkanes are used as fuels due to their energy release on combustion.
- Fractional distillation separates crude oil into fractions.
- Cracking breaks large molecules into smaller ones.
- Reforming rearranges molecules to improve fuel quality.
These processes are essential for producing useful fuels from crude oil.
Example 1:
Write an equation for the complete combustion of ethane.
▶️ Answer/Explanation
\( \mathrm{2C_2H_6 + 7O_2 \rightarrow 4CO_2 + 6H_2O} \)
Example 2:
Explain why cracking is important in the petroleum industry.
▶️ Answer/Explanation
Cracking converts large, less useful hydrocarbons into smaller, more useful ones.
It increases the supply of fuels like petrol.
It also produces alkenes used in making polymers.