Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.13 Combustion pollutants-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.13 Combustion pollutants- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.13 Combustion pollutants- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.13 know that pollutants, including carbon monoxide, oxides of nitrogen and sulfur, carbon particulates and unburned hydrocarbons, are emitted during the combustion of alkane fuels
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.13 Pollutants from Combustion of Alkanes
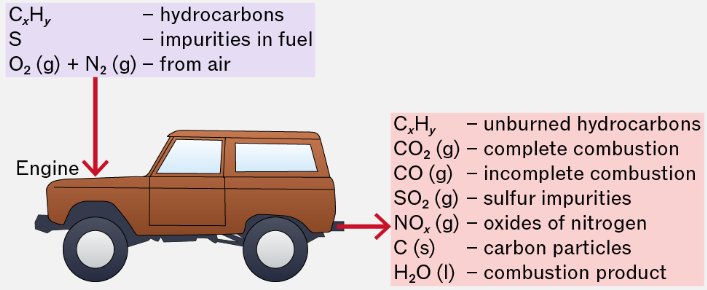
When alkanes are used as fuels, their combustion can produce not only useful energy but also pollutants. These pollutants are harmful to human health and the environment.
Complete vs Incomplete Combustion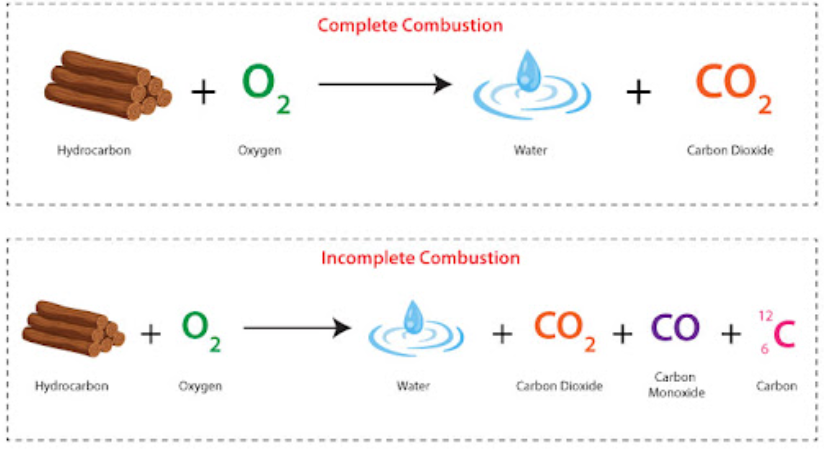
In complete combustion (excess oxygen), alkanes form only carbon dioxide and water:
\( \mathrm{C_{n}H_{2n+2} + O_2 \rightarrow CO_2 + H_2O} \)
However, in real conditions, combustion is often incomplete or occurs at high temperatures, leading to the formation of pollutants.
1. Carbon Monoxide (CO)
Carbon monoxide is a toxic gas formed during incomplete combustion when there is insufficient oxygen.
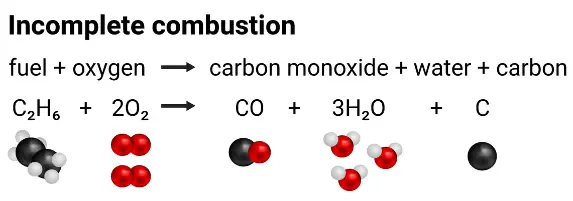
- Colourless and odourless gas.
- Binds strongly to haemoglobin in blood.
- Reduces oxygen transport, potentially causing death.
2. Oxides of Nitrogen (NOₓ)
Oxides of nitrogen (e.g. \( \mathrm{NO} \), \( \mathrm{NO_2} \)) are formed when nitrogen and oxygen in air react at high temperatures inside engines.
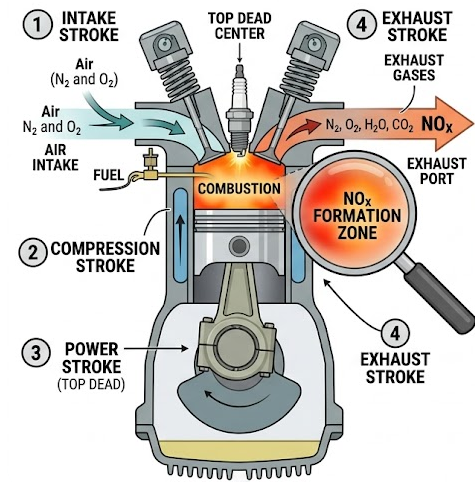
Example: \( \mathrm{N_2 + O_2 \rightarrow 2NO} \)
- Contribute to acid rain.
- Cause respiratory problems.
- Involved in formation of photochemical smog.
3. Sulfur Dioxide (SO₂)
Sulfur dioxide is produced when sulfur impurities in fuels are burned.
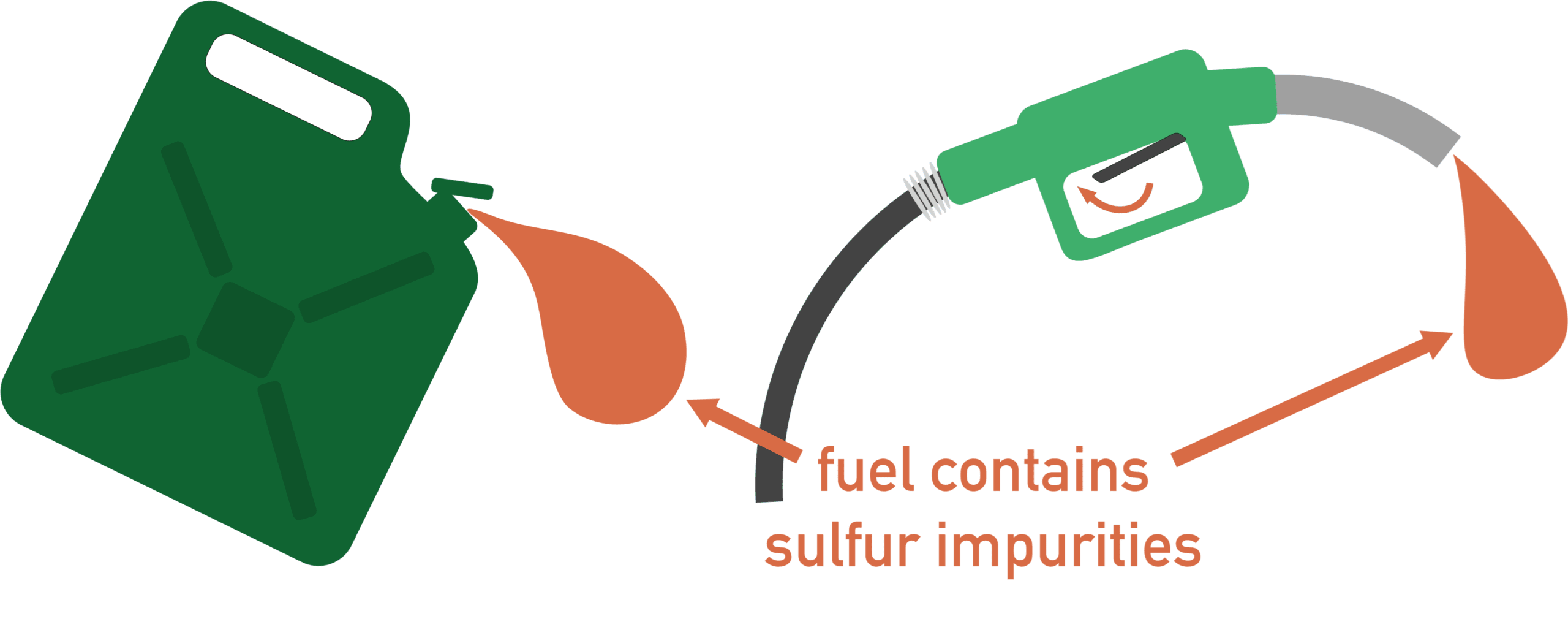
Example: \( \mathrm{S + O_2 \rightarrow SO_2} \)
- Causes acid rain.
- Irritates the respiratory system.
4. Carbon Particulates (Soot)
Carbon particulates are tiny particles of carbon formed during incomplete combustion.
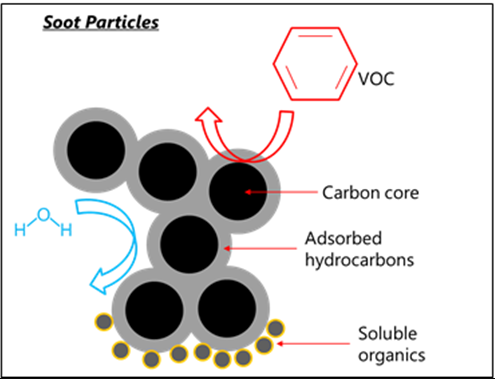
- Produced when there is very limited oxygen.
- Appear as black smoke.
- Can accumulate in lungs and cause health problems.
5. Unburned Hydrocarbons
These are hydrocarbon molecules that escape combustion without reacting.
- Contribute to air pollution.
- React with NOₓ to form photochemical smog.
- Some may be toxic or carcinogenic.
Summary
- Combustion of alkanes produces pollutants under real conditions.
- CO → toxic, reduces oxygen transport.
- NOₓ → acid rain and smog.
- SO₂ → acid rain.
- Carbon particulates → lung damage.
- Unburned hydrocarbons → pollution and smog formation.
Therefore, combustion of alkane fuels can lead to significant environmental and health problems due to the formation of pollutants.
Example 1:
Explain how carbon monoxide is formed during combustion.
▶️ Answer/Explanation
Carbon monoxide is formed when there is insufficient oxygen.
The fuel is not fully oxidised to carbon dioxide.
Therefore, CO is produced instead of CO₂.
Example 2:
Explain why oxides of nitrogen are formed in car engines.
▶️ Answer/Explanation
High temperatures in engines cause nitrogen and oxygen in air to react.
This forms nitrogen oxides such as NO.
Therefore, NOₓ pollutants are produced during combustion.
