Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.14 Environmental impact of combustion-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.14 Environmental impact of combustion- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.14 Environmental impact of combustion- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.14 understand the problems arising from pollutants from the combustion of alkane fuels, limited to the toxicity of carbon monoxide and why it is toxic, and the acidity of oxides of nitrogen and sulfur
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.14 Problems Arising from Pollutants of Alkane Combustion
The combustion of alkane fuels produces pollutants that can cause serious health and environmental problems. At IAL level, focus is on the toxicity of carbon monoxide and the acidic nature of oxides of nitrogen and sulfur.
1. Toxicity of Carbon Monoxide (CO)
Carbon monoxide is a highly toxic, colourless and odourless gas produced during incomplete combustion of alkanes.
Why Carbon Monoxide is Toxic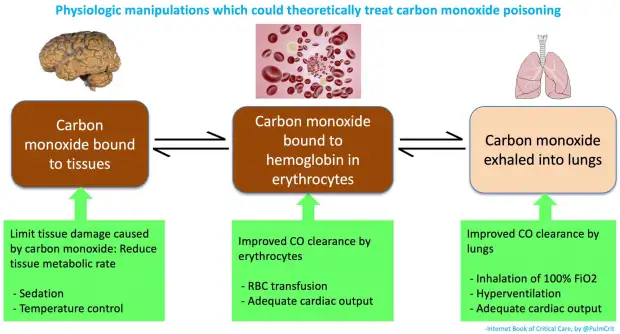
- CO binds strongly to haemoglobin in red blood cells.
- It forms carboxyhaemoglobin.
- This reduces the ability of blood to carry oxygen.
- Body tissues become deprived of oxygen (hypoxia).
Consequences
- Headaches, dizziness and nausea.
- Loss of consciousness.
- Can be fatal in high concentrations.
Therefore, carbon monoxide is dangerous because it prevents oxygen transport in the body.
2. Acidity of Oxides of Nitrogen (NOₓ)
Oxides of nitrogen such as \( \mathrm{NO} \) and \( \mathrm{NO_2} \) are formed at high temperatures during combustion.
Formation of Acids 
- These gases dissolve in water in the atmosphere.
- They form nitric acid:
\( \mathrm{4NO_2 + 2H_2O + O_2 \rightarrow 4HNO_3} \)
Effects
- Cause acid rain.
- Damage plants and aquatic life.
- Corrode buildings and monuments.
Therefore, oxides of nitrogen are harmful because they form acidic solutions in the atmosphere.
3. Acidity of Sulfur Dioxide (SO₂)
Sulfur dioxide is produced when sulfur impurities in fuels are burned.
Formation of Acids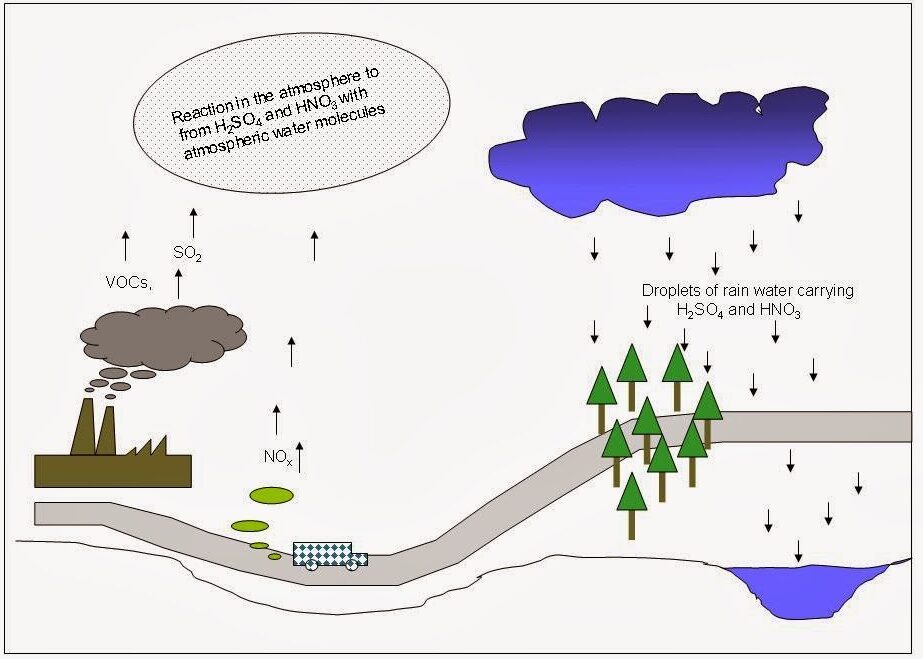
- Dissolves in water to form sulfurous acid:
\( \mathrm{SO_2 + H_2O \rightarrow H_2SO_3} \)
- Can be further oxidised to sulfuric acid:
\( \mathrm{2SO_2 + O_2 \rightarrow 2SO_3} \)
\( \mathrm{SO_3 + H_2O \rightarrow H_2SO_4} \)
Effects
- Causes acid rain.
- Damages ecosystems.
- Corrodes metals and stone structures.
Therefore, sulfur dioxide contributes to environmental damage due to acid formation.
Summary
- CO is toxic because it binds to haemoglobin and reduces oxygen transport.
- NOₓ forms nitric acid, leading to acid rain.
- SO₂ forms sulfurous and sulfuric acids, also causing acid rain.
- These pollutants harm both human health and the environment.
Therefore, pollutants from alkane combustion have significant harmful effects due to toxicity and acidity.
Example 1:
Explain why carbon monoxide is more dangerous than carbon dioxide.
▶️ Answer/Explanation
Carbon monoxide binds strongly to haemoglobin.
This prevents oxygen transport in the blood.
Carbon dioxide does not bind in this way.
Therefore, CO is far more toxic.
Example 2:
Explain how sulfur dioxide leads to acid rain.
▶️ Answer/Explanation
Sulfur dioxide dissolves in water to form sulfurous acid.
It can be further oxidised to sulfuric acid.
These acids fall as acid rain.
Therefore, SO₂ contributes to acid rain formation.
