Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.15 Sustainability and alternative fuels-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.15 Sustainability and alternative fuels- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.15 Sustainability and alternative fuels- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.15 be able to discuss the reasons for developing alternative fuels in terms of sustainability and reducing emissions, including the emission of CO2 and its relationship to climate change
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.15 Development of Alternative Fuels
There is increasing need to develop alternative fuels due to concerns about sustainability and environmental impact of fossil fuels such as alkanes.
1. Sustainability
Fossil fuels are finite (non-renewable) resources formed over millions of years.
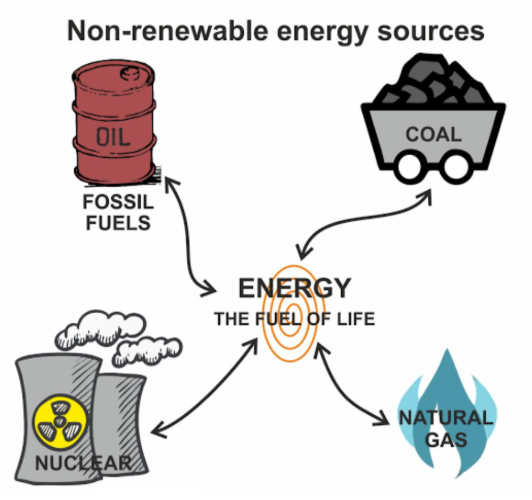
- Supplies are limited and will eventually run out.
- Increasing demand leads to faster depletion.
- Extraction becomes more difficult and expensive over time.
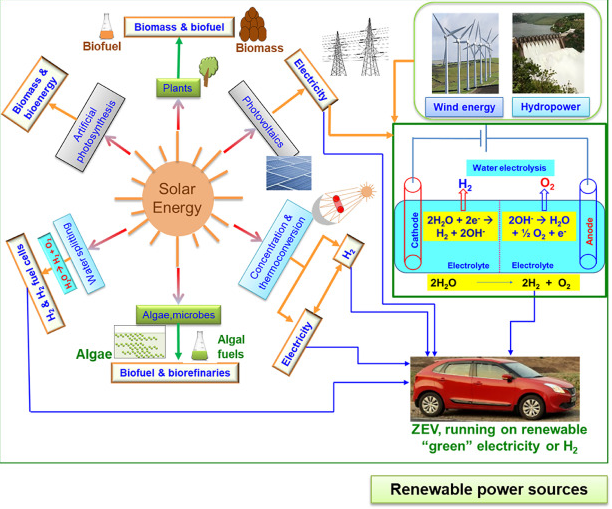
Alternative fuels aim to be renewable, meaning they can be replenished naturally.

- Examples include biofuels and hydrogen.
- Provide long-term energy supply.
Therefore, alternative fuels are developed to ensure a sustainable energy future.
2. Reducing Carbon Dioxide Emissions
Combustion of alkanes produces carbon dioxide, a greenhouse gas:
\( \mathrm{C_{n}H_{2n+2} + O_2 \rightarrow CO_2 + H_2O} \)
- Increased CO₂ levels in the atmosphere enhance the greenhouse effect.
- This leads to global warming and climate change.
Greenhouse Effect
The greenhouse effect is the process by which certain gases in the atmosphere trap heat, leading to an increase in Earth’s temperature. Carbon dioxide (\( \mathrm{CO_2} \)) is one of the most important greenhouse gases.
Step-by-Step Mechanism
1. Incoming Solar Radiation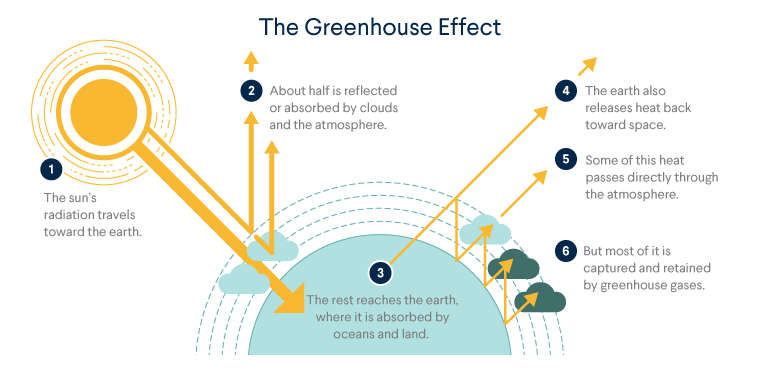
- The Sun emits short-wave radiation (mainly visible light and UV).
- This radiation passes through the atmosphere and reaches the Earth’s surface.
2. Absorption by Earth
- The Earth’s surface absorbs this energy.
- As a result, the surface warms up.
3. Re-emission as Infrared Radiation
- The Earth re-emits energy as long-wave infrared radiation.
- This radiation would normally escape into space.
4. Absorption by Greenhouse Gases
- Greenhouse gases such as \( \mathrm{CO_2} \), \( \mathrm{CH_4} \), and water vapour absorb infrared radiation.
- The energy excites molecular vibrations (bond vibrations increase).
5. Re-radiation of Heat
- The absorbed energy is re-radiated in all directions.
- Some of this heat is sent back toward the Earth’s surface.
6. Net Effect
- Heat is retained in the atmosphere instead of escaping into space.
- This leads to an increase in global average temperature.
Therefore, reducing CO₂ emissions is essential to limit climate change.
3. How Alternative Fuels Reduce Emissions
Biofuels:
- Plants absorb CO₂ during photosynthesis.

- When burned, CO₂ released is roughly balanced (carbon-neutral).
Hydrogen fuel:
- Burns to produce only water: \( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
- No CO₂ emissions during use.
Cleaner combustion:
- Some fuels produce fewer pollutants (e.g. less CO, NOₓ, particulates).
4. Other Environmental Benefits
- Reduced air pollution.
- Lower contribution to acid rain and smog.
- Improved human health.
Summary
- Fossil fuels are non-renewable and unsustainable.
- Their combustion releases CO₂, contributing to climate change.
- Alternative fuels aim to be renewable and reduce emissions.
- They help limit global warming and environmental damage.
Therefore, alternative fuels are developed to provide sustainable energy and reduce the environmental impact of fuel use.
Example 1:
Explain why carbon dioxide contributes to climate change.
▶️ Answer/Explanation
Carbon dioxide absorbs infrared radiation from the Earth.
This traps heat in the atmosphere.
As CO₂ concentration increases, more heat is retained.
Therefore, global temperatures rise, causing climate change.
Example 2:
Explain why hydrogen is considered a clean fuel.
▶️ Answer/Explanation
Hydrogen burns to form only water.
It does not produce carbon dioxide.
Therefore, it does not contribute to greenhouse gas emissions.