Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.16 Carbon neutrality-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.16 Carbon neutrality- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.16 Carbon neutrality- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.16 be able to apply the concept of carbon neutrality to different fuels, such as petrol, bioethanol and hydrogen
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.16 Carbon Neutrality and Different Fuels
The concept of carbon neutrality is important when comparing the environmental impact of different fuels such as petrol, bioethanol and hydrogen.
Definition of Carbon Neutrality
A fuel is carbon neutral if the net carbon dioxide emissions released during its use are equal to the amount of carbon dioxide absorbed from the atmosphere.
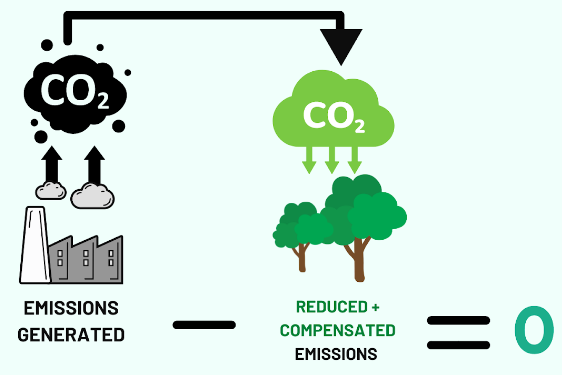
This means there is no overall increase in atmospheric CO₂ levels.
1. Petrol (Fossil Fuel)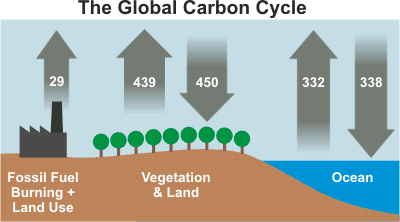
Petrol is a mixture of alkanes obtained from crude oil and is not carbon neutral.
Combustion Equation
\( \mathrm{C_{n}H_{2n+2} + O_2 \rightarrow CO_2 + H_2O} \)
- Releases CO₂ that was stored underground for millions of years.
- No corresponding process removes this CO₂ quickly.
- Leads to a net increase in atmospheric CO₂.
Therefore, petrol contributes significantly to climate change.
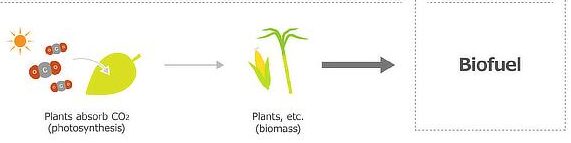
2. Bioethanol (Biofuel)
Bioethanol is produced from plant materials and is considered approximately carbon neutral.
Combustion Equation
\( \mathrm{C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O} \)
Carbon Cycle Explanation
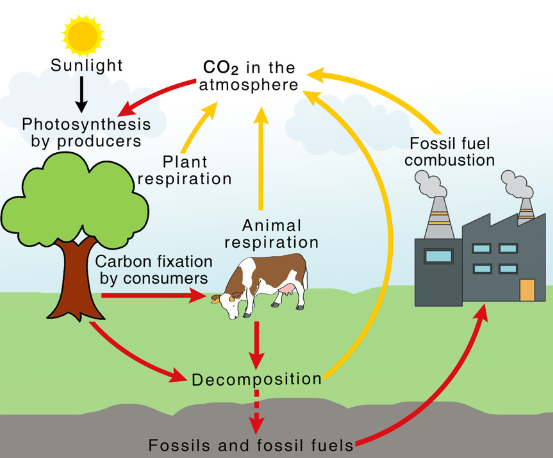
- Plants absorb CO₂ during photosynthesis.
- This carbon is stored in biomass.
- When bioethanol is burned, the same CO₂ is released.
Therefore, there is no net increase in CO₂ if the cycle is balanced.
Limitations
- Energy used in farming, transport and processing may release CO₂.
- Therefore, it is not perfectly carbon neutral in practice.
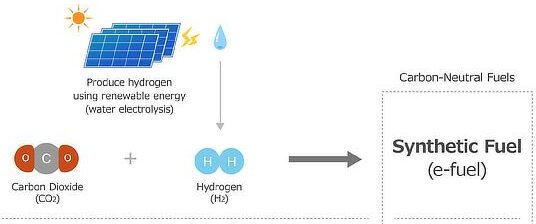
3. Hydrogen
Hydrogen is considered a clean fuel and produces no carbon dioxide when burned.
Combustion Equation
\( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
- Only water is produced.
- No CO₂ emissions during use.
However
- Hydrogen production may involve fossil fuels (e.g. steam reforming).
- If produced using renewable energy (electrolysis), it can be fully sustainable.
Therefore, hydrogen can be carbon neutral depending on how it is produced.
Comparison of Fuels
- Petrol → not carbon neutral, increases CO₂ levels.
- Bioethanol → approximately carbon neutral.
- Hydrogen → no CO₂ on combustion, potentially carbon neutral.
Summary
- Carbon neutrality means no net increase in atmospheric CO₂.
- Fossil fuels are not carbon neutral.
- Biofuels can be nearly carbon neutral due to the carbon cycle.
- Hydrogen produces no CO₂ but depends on production method.
Therefore, evaluating carbon neutrality helps compare the environmental impact of different fuels.
Example 1:
Explain why bioethanol is considered carbon neutral.
▶️ Answer/Explanation
Plants absorb CO₂ during photosynthesis.
This carbon is released again when bioethanol is burned.
Therefore, there is no net increase in atmospheric CO₂.
Example 2:
Explain why hydrogen is not always carbon neutral.
▶️ Answer/Explanation
Hydrogen may be produced using fossil fuels, releasing CO₂.
Therefore, overall emissions may not be zero.
It is only carbon neutral if produced using renewable energy.
