Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.18 Free radical substitution mechanism-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.18 Free radical substitution mechanism- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.18 Free radical substitution mechanism- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.18 understand the mechanism of the free radical substitution reaction between an alkane and a halogen:
i using free radicals, which are species with an unpaired electron, represented by a single dot
ii showing the initiation step of the mechanism, with curly half-arrows for free radical formation
iii showing the propagation and termination steps of the mechanism
iv having limited use in synthesis because of further substitution reactions
Further suggested practical:
Cracking alkanes by thermal decomposition, including liquid paraffin using aluminium oxide as a catalyst
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.18 Free Radical Substitution Mechanism (Alkanes + Halogens)
The reaction between an alkane and a halogen (e.g. \( \mathrm{Cl_2} \), \( \mathrm{Br_2} \)) proceeds via a free radical substitution mechanism. This is a chain reaction involving highly reactive free radicals.
(i) Free Radicals
Free radicals are key species involved in the mechanism of free radical substitution reactions.
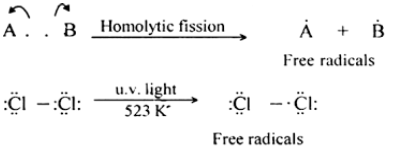
A free radical is a species with an unpaired electron.
- Represented using a dot (•), e.g. \( \mathrm{Cl\cdot} \), \( \mathrm{CH_3\cdot} \).
- Formed by homolytic bond fission.
- Highly reactive due to the presence of an unpaired electron.
- Tend to react quickly to form stable molecules.
Therefore, free radicals are unstable intermediates that drive chain reactions.
(ii) Initiation Step (with Curly Half-Arrows)
The initiation step is the first step in the free radical substitution mechanism, where free radicals are generated.
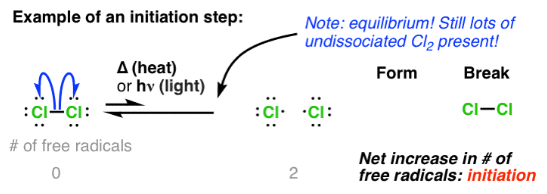
It involves homolytic fission of a halogen molecule using UV light.
\( \mathrm{Cl_2 \xrightarrow{UV} 2Cl\cdot} \)
- The \( \mathrm{Cl–Cl} \) bond breaks evenly.
- Each chlorine atom takes one electron from the bond.
- Two chlorine free radicals are formed.
Curly Half-Arrows
- Used to show movement of a single electron.
- Each arrow starts from the bond and goes to each atom.
- Indicates homolytic bond breaking.
Therefore, the initiation step produces free radicals and starts the chain reaction.
(iii) Propagation and Termination Steps
After initiation, the reaction continues through propagation and eventually stops through termination. These steps explain how the chain reaction proceeds and ends.
Propagation Steps
In propagation, free radicals react with molecules to form new radicals, allowing the reaction to continue.
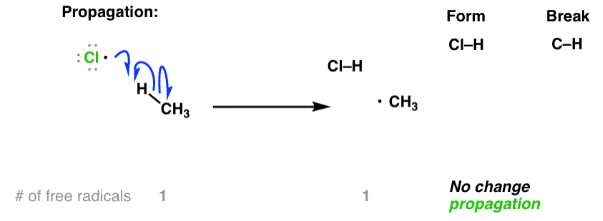
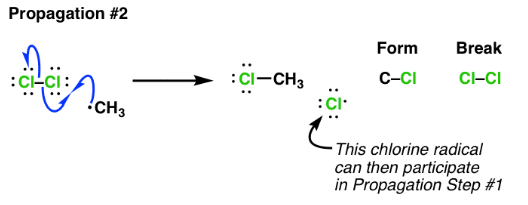
Example (chlorination of methane):
\( \mathrm{Cl\cdot + CH_4 \rightarrow HCl + CH_3\cdot} \)
\( \mathrm{CH_3\cdot + Cl_2 \rightarrow CH_3Cl + Cl\cdot} \)
- A radical reacts with a stable molecule.
- A new radical is formed.
- The chain reaction continues.
- Curly half-arrows are used to show movement of single electrons.
Therefore, propagation sustains the reaction by regenerating radicals.
Termination Steps
In termination, two free radicals combine to form a stable molecule, removing radicals from the system.
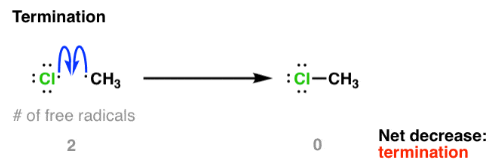
Examples:
\( \mathrm{Cl\cdot + Cl\cdot \rightarrow Cl_2} \)
\( \mathrm{CH_3\cdot + Cl\cdot \rightarrow CH_3Cl} \)
\( \mathrm{CH_3\cdot + CH_3\cdot \rightarrow C_2H_6} \)
- No new radicals are formed.
- The chain reaction slows and stops.
Therefore, termination ends the reaction by removing free radicals.
(iv) Limited Use in Synthesis
Free radical substitution reactions have limited use in synthesis because they produce mixtures of products.
Reason: Further Substitution
- After the first substitution, the product can react further.
- More hydrogen atoms can be replaced by halogen atoms.
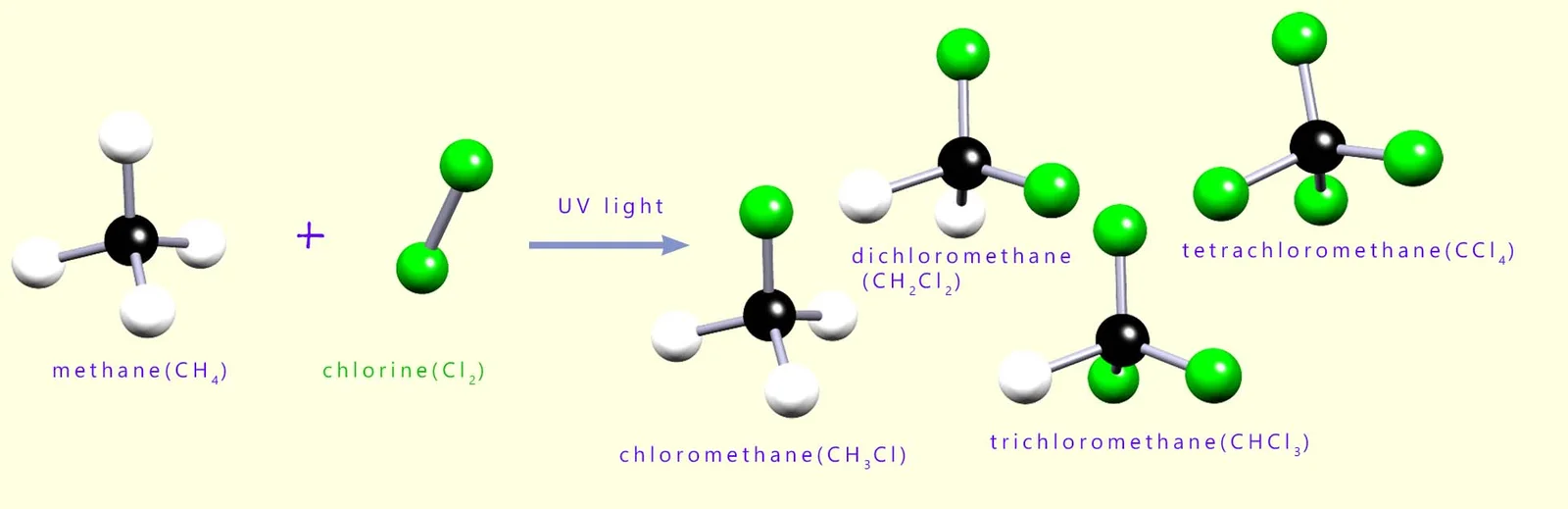
- A mixture of products is formed.
- Difficult to control the extent of substitution.
- Hard to obtain a single pure product.
Therefore, this reaction is not ideal when a specific product is required.
Example 1:
The reaction between methane and chlorine proceeds via a free radical substitution mechanism.
Write equations for the initiation, propagation and one termination step.
▶️ Answer/Explanation
Initiation:
\( \mathrm{Cl_2 \xrightarrow{UV} 2Cl\cdot} \)
Propagation:
\( \mathrm{Cl\cdot + CH_4 \rightarrow HCl + CH_3\cdot} \)
\( \mathrm{CH_3\cdot + Cl_2 \rightarrow CH_3Cl + Cl\cdot} \)
Termination (any one):
\( \mathrm{Cl\cdot + Cl\cdot \rightarrow Cl_2} \)
Free radicals are generated in initiation, regenerated in propagation, and removed in termination.
Example 2:
Explain why the chlorination of methane has limited use in synthesis.
▶️ Answer/Explanation
After the first substitution, the product can undergo further substitution reactions.
\( \mathrm{CH_4 \rightarrow CH_3Cl \rightarrow CH_2Cl_2 \rightarrow CHCl_3 \rightarrow CCl_4} \)
This leads to a mixture of products.
The reaction is difficult to control and does not produce a single desired product in high yield.
Therefore, free radical substitution has limited use in synthesis.
Further Suggested Practical: Cracking of Alkanes (Thermal Decomposition)
This practical demonstrates the cracking of long-chain alkanes into smaller, more useful hydrocarbons using heat and a catalyst such as aluminium oxide.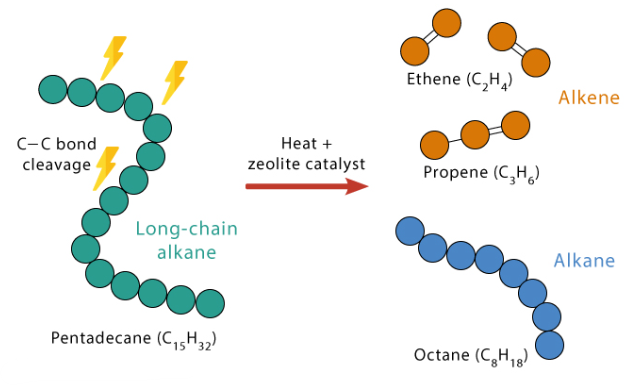
Aim
To crack a long-chain alkane (liquid paraffin) into smaller alkanes and alkenes and test the products formed.
Principle
Cracking is the thermal decomposition of large hydrocarbon molecules into smaller molecules in the presence of a catalyst.
General reaction:
\( \mathrm{C_{n}H_{2n+2} \rightarrow smaller\ alkane + alkene} \)
Example:
- \( \mathrm{C_{10}H_{22} \rightarrow C_8H_{18} + C_2H_4} \)
Apparatus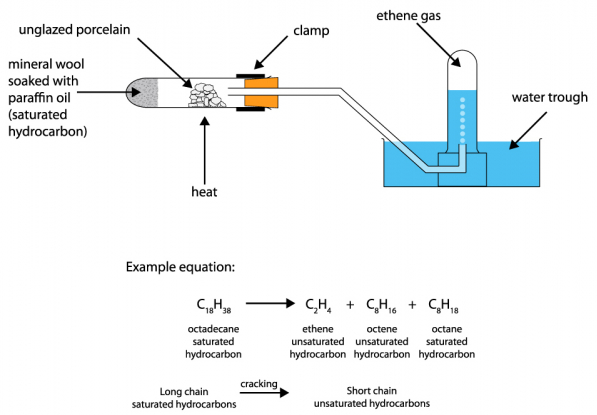
- Hard glass test tube
- Liquid paraffin
- Aluminium oxide (catalyst)
- Mineral wool
- Delivery tube
- Bunsen burner
- Bromine water
Procedure
- Soak mineral wool with liquid paraffin and place it at one end of the test tube.
- Place aluminium oxide in the middle of the tube.
- Heat the aluminium oxide strongly first.
- Then gently heat the paraffin.
- Vapours pass over the hot catalyst and crack into smaller molecules.
- Collect the gaseous products.
- Test gases with bromine water.
Observations
- Gas is produced.
- Bromine water changes from orange to colourless.
This indicates the presence of alkenes.
Explanation
- Large alkane molecules break into smaller molecules.
- Some products are alkanes (saturated).
- Some are alkenes (unsaturated).
- Alkenes decolourise bromine water.
Safety Precautions
- Use a fume cupboard (hydrocarbon vapours are flammable).
- Keep apparatus pointed away from people.
- Avoid inhaling gases.
- Handle hot apparatus with care.
Conclusion
- Cracking produces smaller hydrocarbons from larger ones.
- Alkenes are formed, confirmed by bromine water test.
- Aluminium oxide acts as a catalyst.
Therefore, cracking is an important industrial and laboratory process for producing useful fuels and alkenes.
Example 1:
Explain why aluminium oxide is used in this experiment.
▶️ Answer/Explanation
Aluminium oxide acts as a catalyst.
It lowers the activation energy of the cracking reaction.
Therefore, cracking occurs more efficiently.
Example 2:
Explain how you would test that alkenes are formed.
▶️ Answer/Explanation
Bubble the gas through bromine water.
If alkenes are present, bromine water decolourises.
Therefore, this confirms the formation of unsaturated hydrocarbons.
