Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.4 Homologous series and functional groups-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.4 Homologous series and functional groups- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.4 Homologous series and functional groups- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.4 understand the concepts of homologous series and functional group
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.4 Homologous Series and Functional Group
Organic chemistry involves studying groups of compounds with similar structures and properties. Two key concepts used to classify and understand these compounds are homologous series and functional groups.
Homologous Series
A homologous series is a family of organic compounds with the same functional group and similar chemical properties.
Members of a homologous series follow a general pattern and differ from each other by a repeating unit.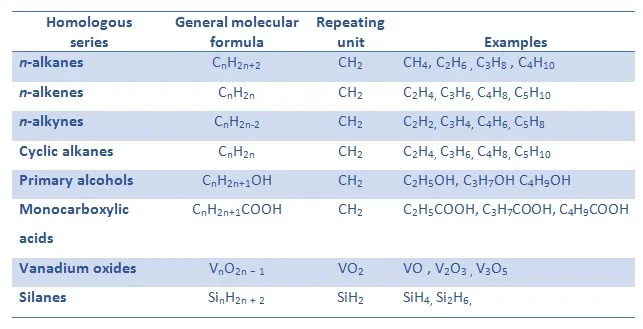
Key Characteristics
- All members have the same functional group.
- They have the same general formula.
- Successive members differ by a \( \mathrm{CH_2} \) unit.
- They show similar chemical properties.
- They show a gradual change in physical properties (e.g. boiling point increases with chain length).
Therefore, a homologous series allows systematic study of related organic compounds.
Examples of Homologous Series
- Alkanes: \( \mathrm{C_nH_{2n+2}} \)
- Alkenes: \( \mathrm{C_nH_{2n}} \)
- Alcohols: \( \mathrm{C_nH_{2n+1}OH} \)
Therefore, both concepts work together to explain patterns in organic chemistry.
Example 1:
Explain why members of a homologous series have similar chemical properties.
▶️ Answer/Explanation
All members of a homologous series have the same functional group.
The functional group determines the type of chemical reactions.
Therefore, all members undergo similar reactions.
Example 2:
Explain why boiling points increase along a homologous series of alkanes.
▶️ Answer/Explanation
As the chain length increases, the number of electrons increases.
This strengthens intermolecular forces (London forces).
More energy is required to overcome these forces.
Therefore, boiling points increase.
Functional Group
A functional group is an atom or group of atoms responsible for the characteristic chemical reactions of an organic compound.
The functional group is the reactive part of the molecule, and it determines the chemical behaviour of the compound, regardless of the length or structure of the carbon chain.Compounds that contain the same functional group form a homologous series and undergo similar types of reactions.
Key Features of Functional Groups
- Determine the type of chemical reactions a compound undergoes.
- Responsible for reactivity and chemical properties.
- Remain unchanged in many reactions involving other parts of the molecule.
- Allow classification of organic compounds into families.
Common Functional Groups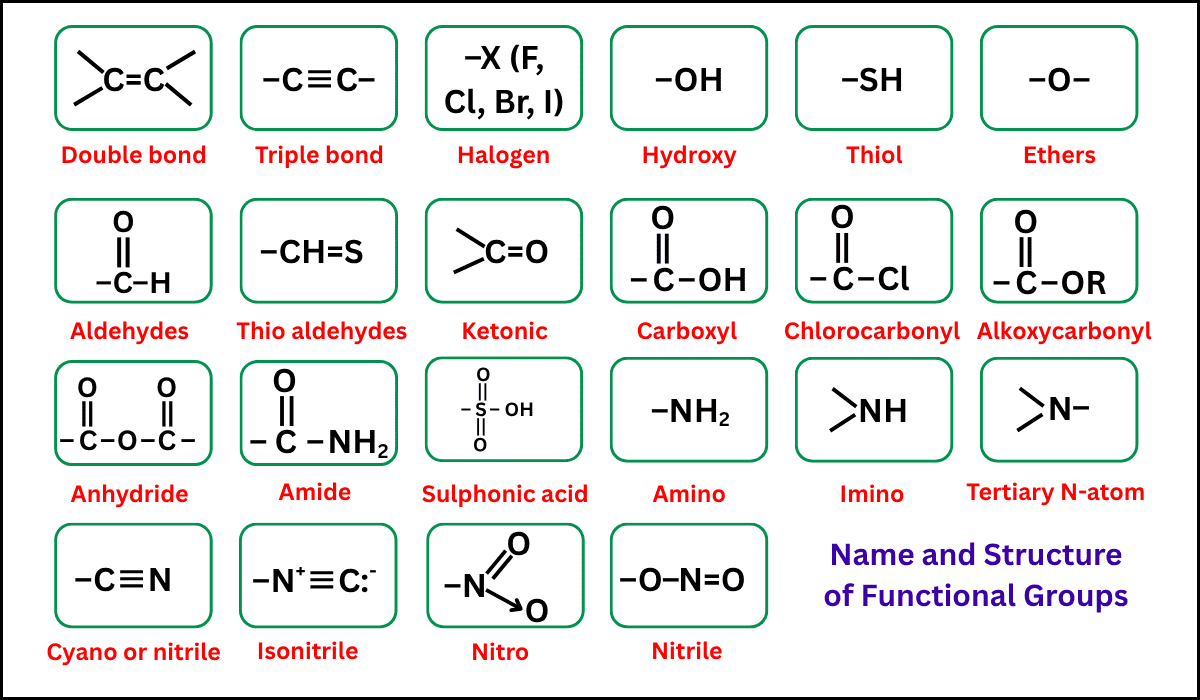
1. Alkane
- Functional group: none (only C–C single bonds)
- General formula: \( \mathrm{C_nH_{2n+2}} \)
- Saturated hydrocarbons
- Relatively unreactive (except combustion and substitution)
2. Alkene
- Functional group: C=C double bond
- General formula: \( \mathrm{C_nH_{2n}} \)
- Unsaturated hydrocarbons
- Undergo addition reactions
3. Haloalkane
- Functional group: C–X (X = halogen: Cl, Br, I)
- Formed by substitution reactions of alkanes or addition to alkenes
- Undergo nucleophilic substitution reactions
4. Alcohol
- Functional group: –OH (hydroxyl group)
- General formula: \( \mathrm{C_nH_{2n+1}OH} \)
- Can form hydrogen bonds
- Undergo oxidation and substitution reactions
5. Aldehyde
- Functional group: –CHO
- Contains carbonyl group at end of chain
- Easily oxidised to carboxylic acids
6. Ketone
- Functional group: –CO– (carbonyl in middle)
- More resistant to oxidation than aldehydes
7. Carboxylic Acid
- Functional group: –COOH
- Weak acids
- Undergo neutralisation and esterification reactions
8. Ester
- Functional group: –COO–
- Formed from carboxylic acids and alcohols
- Often have characteristic fruity smells
9. Amine
- Functional group: –NH₂ (or substituted forms)
- Basic in nature
- React with acids to form ammonium salts
10. Amide
- Functional group: –CONH₂
- Derived from carboxylic acids
- Found in proteins (peptide bonds)
Importance of Functional Groups
- Allow prediction of chemical reactions.
- Help classify organic compounds systematically.
- Determine physical properties such as boiling point and solubility.
Therefore, functional groups are central to understanding organic chemistry at IAL level, as they link structure to reactivity and properties.
Example 1:
Explain why ethanol and ethanoic acid have very different chemical properties.
▶️ Answer/Explanation
Ethanol contains the –OH functional group, while ethanoic acid contains the –COOH group.
Different functional groups determine different chemical behaviour.
Therefore, the compounds undergo different reactions and have different properties.
Example 2:
Identify the functional group in propene and describe its reactivity.
▶️ Answer/Explanation
Propene contains a C=C double bond, which is the alkene functional group.
This bond has high electron density.
Therefore, it readily undergoes addition reactions.
