Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.5 IUPAC naming and formulae-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.5 IUPAC naming and formulae- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.5 IUPAC naming and formulae- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.5 be able to apply the rules of International Union of Pure and Applied Chemistry (IUPAC) nomenclature to:
i name compounds relevant to this specification
ii draw these compounds, as they are encountered in the specification, using structural, displayed and skeletal formulae
Students will be expected to know prefixes for compounds up to C10
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.5 (i) IUPAC Nomenclature – Naming Organic Compounds
The International Union of Pure and Applied Chemistry (IUPAC) system provides a set of rules used to give systematic and unambiguous names to organic compounds. This ensures that each compound has a unique name that reflects its structure.
Basic Principle
The name of an organic compound is based on its longest carbon chain, the functional group, and any substituents attached to the chain.
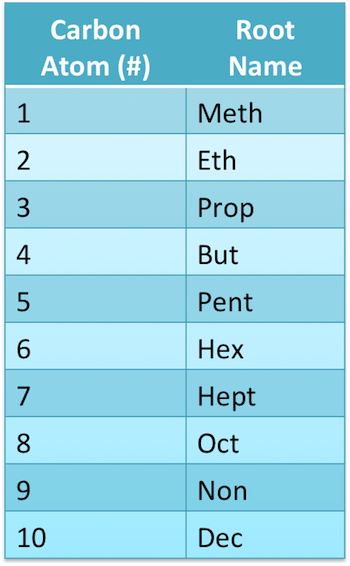
Prefixes for Number of Carbon Atoms (up to C10)
Common Suffixes (Functional Groups)
- Alkane → -ane
- Alkene → -ene
- Alcohol → -ol
- Aldehyde → -al
- Ketone → -one
- Carboxylic acid → -oic acid
Step-by-Step Rules for Naming
1. Identify the Longest Carbon Chain
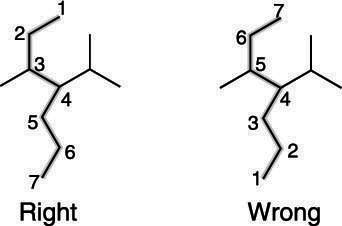
- Find the longest continuous chain of carbon atoms.
- This determines the base name (e.g. methane, ethane, propane).
2. Identify the Functional Group
- The functional group determines the suffix.
- It has priority in numbering.
3. Number the Carbon Chain
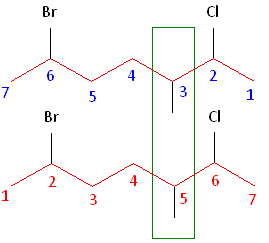
- Number the chain so that the functional group gets the lowest possible number.
- If no functional group, prioritise double/triple bonds or substituents.
4. Identify and Name Substituents
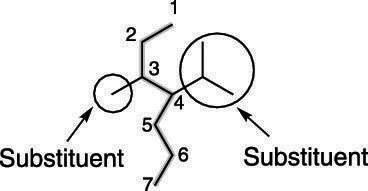
- Substituents are groups attached to the main chain (e.g. methyl, ethyl).
- Their positions are indicated by numbers.
5. Use Multipliers if Needed
- di-, tri-, tetra- for multiple identical substituents.
- Example: 2,2-dimethylpropane
6. Arrange Substituents Alphabetically
- Ignore prefixes like di-, tri- when ordering alphabetically.
7. Write the Full Name
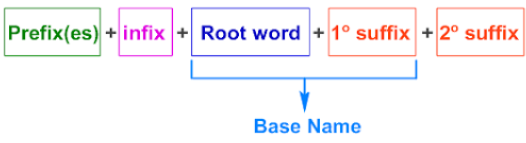
- Format: position–substituent + parent chain + suffix
- Use hyphens (-) between numbers and words.
- Use commas (,) between numbers.
Examples
- \( \mathrm{CH_3CH_2CH_3} \) → propane
- \( \mathrm{CH_3CH(CH_3)CH_3} \) → 2-methylpropane
- \( \mathrm{CH_3CH_2OH} \) → ethanol
Important Notes
- Always give the functional group the lowest number.
- Double bonds must be numbered (e.g. but-1-ene).
- Do not include unnecessary numbers (e.g. ethanol, not ethan-1-ol at this level unless needed).
Therefore, IUPAC nomenclature allows systematic naming of organic compounds based on structure, ensuring clarity and consistency.
Example 1:
Name the compound: \( \mathrm{CH_3CH(CH_3)CH_2CH_3} \).
▶️ Answer/Explanation
Longest chain has 4 carbons → butane.
There is a methyl substituent on carbon 2.
Therefore, the name is 2-methylbutane.
Example 2:
Name the compound: \( \mathrm{CH_2=CHCH_3} \).
▶️ Answer/Explanation
Longest chain has 3 carbons → prop-.
There is a double bond starting at carbon 1.
Therefore, the name is prop-1-ene (or propene).
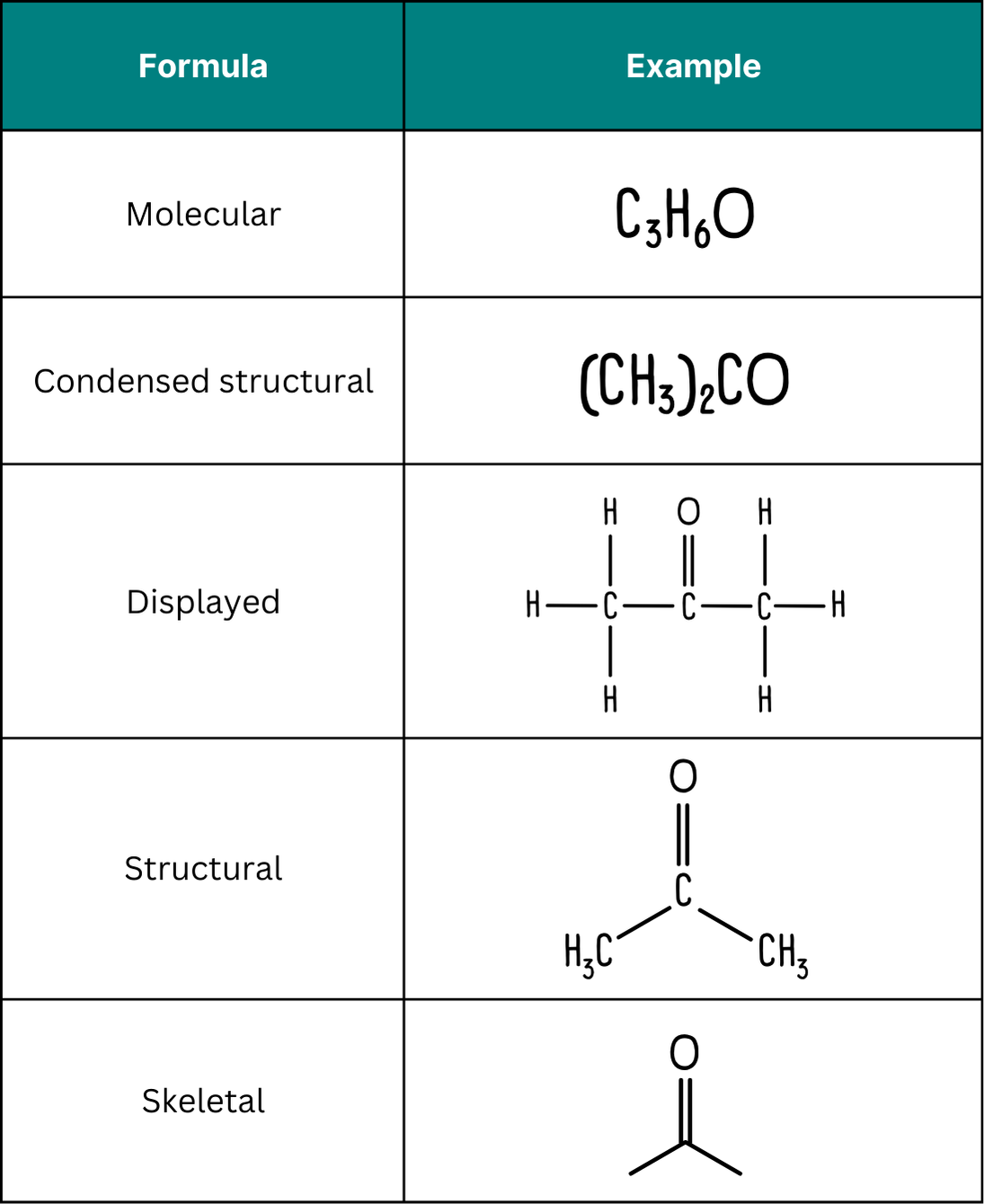
4.5 (ii) Drawing Organic Compounds (Structural, Displayed and Skeletal Formulae)
In IAL chemistry, it is essential to be able to represent organic compounds using different types of formulae. These show varying levels of detail about the arrangement of atoms and bonds.
1. Structural Formula
A structural formula shows how atoms are arranged in a molecule, but does not show all bonds explicitly.
- Atoms are written in sequence (e.g. CH₃CH₂OH).
- Bonds between atoms are implied.
- Quick and convenient representation.
Example
- Ethanol → \( \mathrm{CH_3CH_2OH} \)
2. Displayed Formula
A displayed formula shows all atoms and all bonds in a molecule.
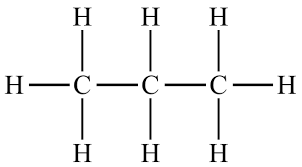
- Every bond is drawn as a line.
- Shows the exact arrangement of atoms.
- Useful for understanding bonding and reactions.
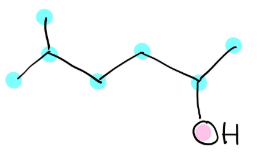
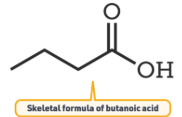
3. Skeletal Formula
A skeletal formula is a simplified representation that shows only the carbon skeleton.
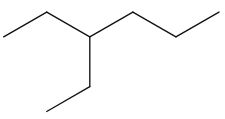
- Carbon atoms are not shown explicitly.
- Hydrogen atoms attached to carbon are not shown.
- Each line represents a bond.
- Ends and corners represent carbon atoms.
- Functional groups are shown explicitly (e.g. –OH, –COOH).
Example
Key Rules for Skeletal Formula
- Each vertex (corner) represents a carbon atom.
- Hydrogens attached to carbon are omitted.
- Heteroatoms (O, N, halogens) must be shown.
- Double and triple bonds are shown with multiple lines.
Comparison of the Three Formulae
- Structural → shows connectivity, compact form.
- Displayed → shows all atoms and bonds clearly.
- Skeletal → simplified, used for larger molecules.
When to Use Each
- Structural → quick writing in exams.
- Displayed → explaining bonding or mechanisms.
- Skeletal → complex organic molecules.
Therefore, being able to draw and interpret all three forms is essential for understanding and communicating organic structures.
Example 1:
Draw the displayed formula of propene.
▶️ Answer/Explanation
Propene has three carbon atoms with a double bond.
Displayed formula shows all bonds:
H₂C=CH–CH₃ (with all bonds drawn explicitly).
Example 2:
Explain why skeletal formulae do not show hydrogen atoms bonded to carbon.
▶️ Answer/Explanation
Each carbon atom forms four bonds.
The number of hydrogen atoms can be inferred from this.
Therefore, they are omitted to simplify the diagram.