Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.6 Classification of reactions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.6 Classification of reactions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.6 Classification of reactions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.6 be able to classify reactions as addition, substitution, oxidation, reduction or polymerisation
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.6 Classification of Organic Reactions
Organic reactions can be classified into different types based on what happens to the molecules during the reaction. The main types required at IAL level are addition, substitution, oxidation, reduction and polymerisation.
Addition Reactions
An addition reaction is one in which two molecules combine to form a single product, usually involving the breaking of a multiple bond.
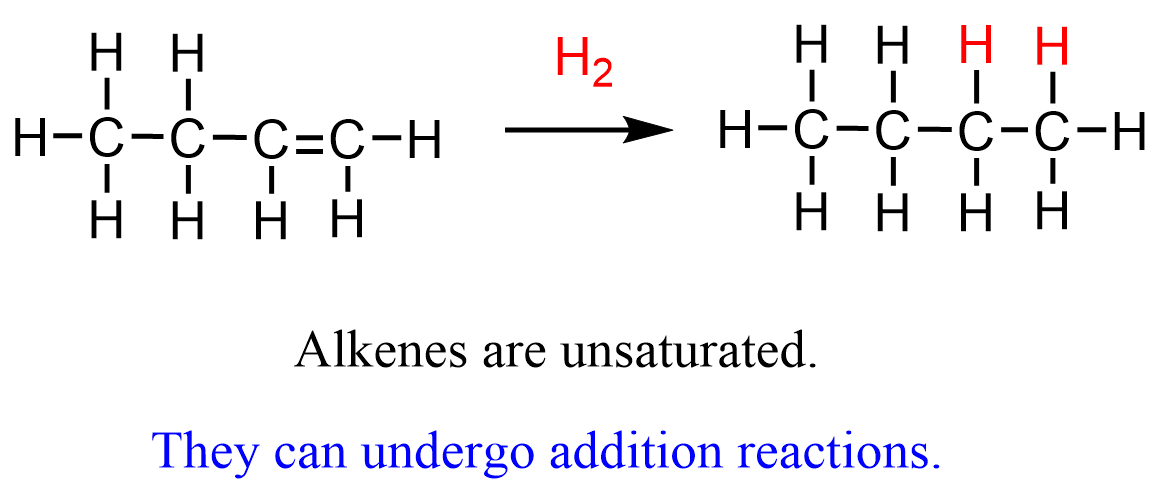
- Occurs mainly in alkenes (C=C double bonds).
- The double bond breaks and atoms are added across it.
- Product is more saturated than reactant.
Examples
- Hydrogenation: \( \mathrm{C_2H_4 + H_2 \rightarrow C_2H_6} \)
- Halogenation: addition of Br₂ to alkenes
Therefore, addition reactions convert unsaturated compounds into saturated ones.
Substitution Reactions
A substitution reaction is one in which one atom or group is replaced by another.
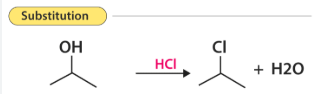
- Common in alkanes and haloalkanes.
- One bond is broken and a new bond is formed.
Examples
- Halogenation of methane: \( \mathrm{CH_4 + Cl_2 \rightarrow CH_3Cl + HCl} \)
- Hydrolysis of haloalkanes: replacing halogen with OH⁻
Therefore, substitution reactions involve replacement without changing saturation significantly.
Oxidation Reactions
Oxidation in organic chemistry involves gain of oxygen, loss of hydrogen, or increase in oxidation state.
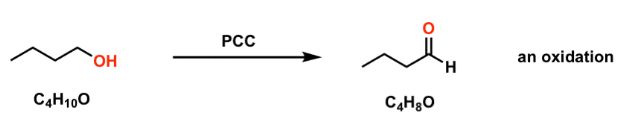
- Common for alcohols and aldehydes.
- Often uses oxidising agents (e.g. acidified potassium dichromate).
Examples
- Primary alcohol → aldehyde → carboxylic acid
- Secondary alcohol → ketone
Therefore, oxidation increases the oxygen content or reduces hydrogen content.
Reduction Reactions
Reduction is the opposite of oxidation and involves gain of hydrogen, loss of oxygen, or decrease in oxidation state.

- Often occurs alongside oxidation (redox reactions).
- Common in converting carbonyl compounds to alcohols.
Examples
- Aldehyde → primary alcohol
- Ketone → secondary alcohol
- Alkene + H₂ → alkane (also addition + reduction)
Therefore, reduction increases hydrogen content or decreases oxygen content.
Polymerisation Reactions
Polymerisation is a reaction in which many small molecules (monomers) join together to form a large molecule (polymer).
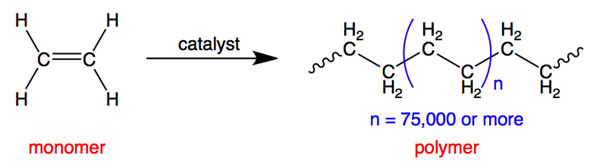
- Occurs mainly with alkenes (addition polymerisation).
- Double bonds open and link together repeatedly.
- Produces long-chain macromolecules.
Example
- Ethene → poly(ethene)
Therefore, polymerisation forms large molecules from many repeating units.
Summary of Reaction Types
- Addition → adds atoms across multiple bonds.
- Substitution → replaces one group with another.
- Oxidation → gain of O / loss of H.
- Reduction → gain of H / loss of O.
- Polymerisation → forms large molecules from monomers.
These classifications help in understanding and predicting organic reaction mechanisms and products.
Example 1:
Classify the reaction: \( \mathrm{C_2H_4 + Br_2 \rightarrow C_2H_4Br_2} \).
▶️ Answer/Explanation
Bromine is added across the C=C double bond.
Two molecules combine to form one product.
Therefore, this is an addition reaction.
Example 2:
Classify the reaction where ethanol is converted to ethanoic acid.
▶️ Answer/Explanation
Ethanol gains oxygen and loses hydrogen during the reaction.
This increases its oxidation state.
Therefore, the reaction is oxidation.
