Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.7 Homolytic and heterolytic bond fission-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.7 Homolytic and heterolytic bond fission- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.7 Homolytic and heterolytic bond fission- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.7 understand that bond breaking can be:
i homolytic, to produce free radicals
ii heterolytic, to produce ions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.7 (i) Homolytic Bond Fission
In organic reactions, bonds can break in different ways. One important type is homolytic bond fission, which leads to the formation of highly reactive species called free radicals.
Definition
Homolytic bond fission is the breaking of a covalent bond in which each atom takes one electron from the shared pair.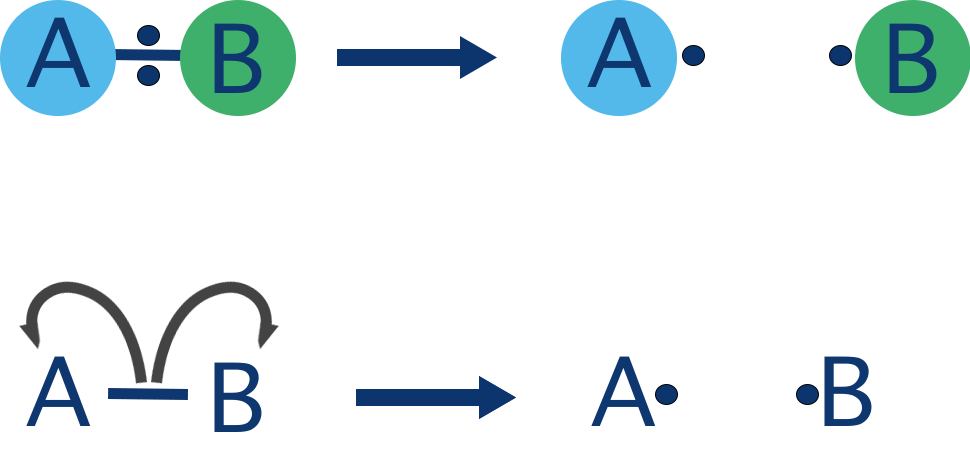
This results in the formation of two species, each with an unpaired electron.
Free Radicals
A free radical is a species with an unpaired electron.
- Highly reactive due to the unpaired electron.
- Represented using a dot (•), e.g. Cl•
- Tend to react quickly to form stable molecules.
Example of Homolytic Fission
\( \mathrm{Cl_2 \rightarrow 2Cl\cdot} \)
- The Cl–Cl bond breaks evenly.
- Each chlorine atom takes one electron.
- Two chlorine radicals are formed.
Conditions for Homolytic Fission
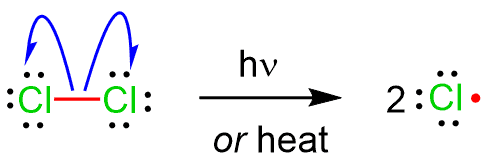
- Requires energy input such as UV light or heat.
- Occurs mainly in non-polar bonds.
- Common in reactions involving halogens.
Mechanism Involvement
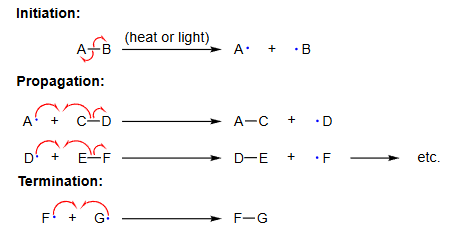
- Homolytic fission is the first step in free radical substitution reactions.
- It leads to chain reactions involving initiation, propagation and termination steps.
Summary
- Bond breaks evenly.
- Each atom gets one electron.
- Forms free radicals.
- Requires energy (UV light or heat).
Therefore, homolytic bond fission produces reactive free radicals and is important in reactions such as free radical substitution.
Example 1:
Explain what happens during homolytic fission of a chlorine molecule.
▶️ Answer/Explanation
The Cl–Cl bond breaks evenly.
Each chlorine atom takes one electron from the bond.
Two chlorine free radicals are formed.
Example 2:
Explain why free radicals are highly reactive.
▶️ Answer/Explanation
Free radicals contain an unpaired electron.
This makes them unstable.
They react quickly to pair up the electron and form stable molecules.
4.7 (ii) Heterolytic Bond Fission
Another way in which covalent bonds can break is by heterolytic bond fission, which leads to the formation of charged species called ions. This type of bond breaking is very important in many organic reaction mechanisms.
Definition
Heterolytic bond fission is the breaking of a covalent bond in which both electrons in the shared pair are taken by one atom.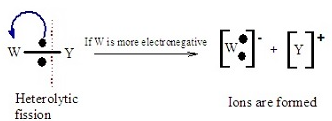
This results in the formation of two oppositely charged ions.
Types of Ions Formed
- Cation: positively charged ion (loss of electrons)
- Anion: negatively charged ion (gain of electrons)
Example of Heterolytic Fission
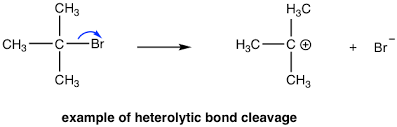
\( \mathrm{C–Br \rightarrow C^+ + Br^-} \)
- Both bonding electrons move to bromine.
- Bromine forms a negative ion (Br⁻).
- Carbon forms a positive ion (C⁺).
Curly Arrow Representation
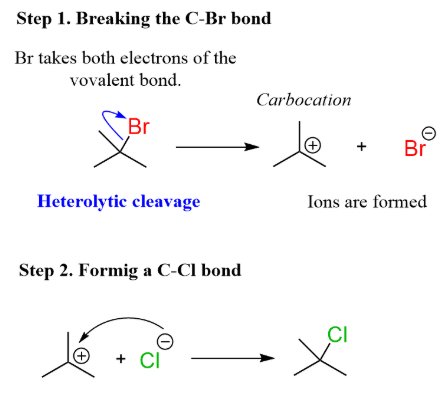
- A curly arrow is used to show movement of an electron pair.
- The arrow starts from the bond and points to the atom gaining both electrons.
Conditions for Heterolytic Fission
- Occurs in polar bonds.
- Favoured when there is a large difference in electronegativity between atoms.
- Often occurs in polar solvents (e.g. water).
Role in Organic Mechanisms
- Leads to formation of electrophiles (electron pair acceptors).
- Leads to formation of nucleophiles (electron pair donors).
- Common in reactions such as nucleophilic substitution and addition.
Summary
- Bond breaks unevenly.
- Both electrons go to one atom.
- Forms ions (cation and anion).
- Occurs in polar bonds and solvents.
Therefore, heterolytic bond fission produces ions and is fundamental in many organic reaction mechanisms involving electrophiles and nucleophiles.
Example 1:
Explain what happens during heterolytic fission of a C–Br bond.
▶️ Answer/Explanation
The bonding pair of electrons moves entirely to the bromine atom.
Bromine becomes Br⁻ (anion).
Carbon loses electrons and becomes C⁺ (cation).
Example 2:
Explain why heterolytic fission is more likely in polar bonds.
▶️ Answer/Explanation
In polar bonds, electrons are already unequally shared.
The more electronegative atom attracts electrons more strongly.
Therefore, it is more likely to take both electrons during bond breaking.
