Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.8 Free radicals and electrophiles-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.8 Free radicals and electrophiles- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.8 Free radicals and electrophiles- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.8 know definitions of the terms ‘free radical’ and ‘electrophile’
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.8 Free Radical and Electrophile
In organic chemistry, different types of reactive species are involved in reaction mechanisms. Two important types are free radicals and electrophiles.
Free Radical
A free radical is a species that contains an unpaired electron.

Free radicals are highly reactive because the unpaired electron makes the species unstable.
- Formed by homolytic bond fission.
- Represented by a dot (•), e.g. \( \mathrm{Cl\cdot} \).
- React rapidly to form stable molecules.
Therefore, a free radical is a highly reactive species due to the presence of an unpaired electron.
Electrophile
An electrophile is a species that accepts a pair of electrons to form a covalent bond.
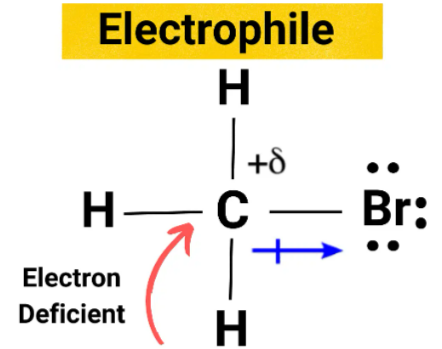
Electrophiles are electron-deficient and are attracted to regions of high electron density.
- Often positively charged or partially positive (δ⁺).
- React with nucleophiles (electron pair donors).
- Examples include \( \mathrm{H^+} \), \( \mathrm{NO_2^+} \), and carbocations.
Therefore, an electrophile is an electron pair acceptor that reacts with electron-rich species.
Key Differences
- Free radical → has an unpaired electron.
- Electrophile → accepts an electron pair.
- Free radicals formed by homolytic fission; electrophiles formed by heterolytic processes.
These species play key roles in different types of organic reaction mechanisms.
Example 1:
Explain why a chlorine atom (Cl•) is a free radical.
▶️ Answer/Explanation
The chlorine atom has an unpaired electron.
This makes it unstable and highly reactive.
Therefore, it is classified as a free radical.
Example 2:
Explain why \( \mathrm{H^+} \) is an electrophile.
▶️ Answer/Explanation
\( \mathrm{H^+} \) has no electrons and is electron-deficient.
It can accept a pair of electrons to form a covalent bond.
Therefore, it acts as an electrophile.
