Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 4.9 General formula; saturated hydrocarbons-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.9 General formula; saturated hydrocarbons- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 4.9 General formula; saturated hydrocarbons- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
4.9 know the general formula of alkanes and cycloalkanes, and understand that they are hydrocarbons (compounds of carbon and hydrogen only) which are saturated (contain single bonds only)
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
4.9 Alkanes and Cycloalkanes
Alkanes and cycloalkanes are important classes of organic compounds known as hydrocarbons. Understanding their structure and general formula is essential in organic chemistry.
Hydrocarbons
Hydrocarbons are compounds that contain only carbon and hydrogen atoms.
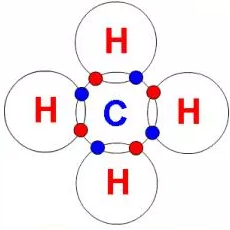
- No other elements are present.
- Found in fuels such as natural gas and petroleum.
Alkanes
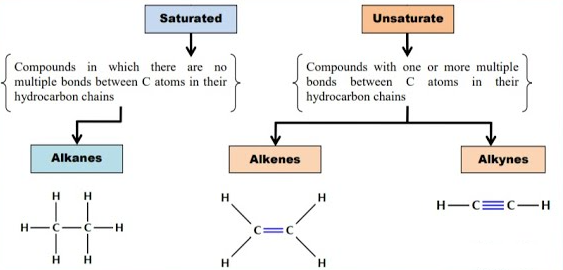
Alkanes are saturated hydrocarbons that contain only single covalent bonds (C–C and C–H).
General Formula of Alkanes
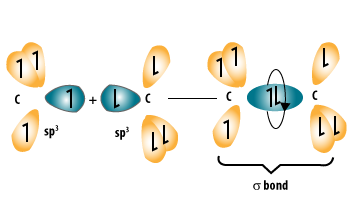
\( \mathrm{C_nH_{2n+2}} \)
- Applies to straight-chain and branched alkanes.
- Each carbon forms four single bonds.
Examples of Alkanes
- Methane → \( \mathrm{CH_4} \)
- Ethane → \( \mathrm{C_2H_6} \)
- Propane → \( \mathrm{C_3H_8} \)
Therefore, alkanes are fully saturated because they contain the maximum number of hydrogen atoms possible.
Cycloalkanes
Cycloalkanes are saturated hydrocarbons with carbon atoms arranged in a ring.
General Formula of Cycloalkanes
\( \mathrm{C_nH_{2n}} \)
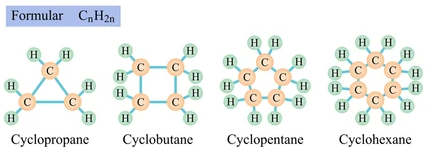
- Two hydrogen atoms are fewer than corresponding alkanes.
- This is because the ends of the chain join to form a ring.
Examples of Cycloalkanes
- Cyclopropane → \( \mathrm{C_3H_6} \)
- Cyclobutane → \( \mathrm{C_4H_8} \)
- Cyclohexane → \( \mathrm{C_6H_{12}} \)
Saturated Nature
Both alkanes and cycloalkanes are saturated, meaning they contain only single bonds and no double or triple bonds.
- Each carbon forms four single covalent bonds.
- They cannot undergo addition reactions easily.
- They are relatively less reactive than unsaturated compounds.
Summary
- Hydrocarbons contain only carbon and hydrogen.
- Alkanes → \( \mathrm{C_nH_{2n+2}} \), open-chain saturated hydrocarbons.
- Cycloalkanes → \( \mathrm{C_nH_{2n}} \), ring structures.
- Both are saturated (only single bonds).
Therefore, alkanes and cycloalkanes are saturated hydrocarbons with distinct structures but similar bonding characteristics.
Example 1:
Explain why cycloalkanes have the formula \( \mathrm{C_nH_{2n}} \) instead of \( \mathrm{C_nH_{2n+2}} \).
▶️ Answer/Explanation
In cycloalkanes, the carbon chain forms a ring.
Two hydrogen atoms are lost when the ends join together.
Therefore, the formula becomes \( \mathrm{C_nH_{2n}} \).
Example 2:
Explain why alkanes are described as saturated hydrocarbons.
▶️ Answer/Explanation
Alkanes contain only single covalent bonds between carbon atoms.
Each carbon is bonded to the maximum number of hydrogen atoms possible.
Therefore, they are saturated with hydrogen.
