Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.1 General formula; C=C (σ and π bonds)-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.1 General formula- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.1 General formula; C=C (σ and π bonds)- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.1 know the general formula of alkenes and understand that alkenes and cycloalkenes are hydrocarbons which are unsaturated (have a carbon–carbon double bond which consists of a σ bond and a π bond)
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.1 Alkenes and Cycloalkenes
Alkenes are an important homologous series of hydrocarbons characterised by the presence of a carbon–carbon double bond.
General Formula of Alkenes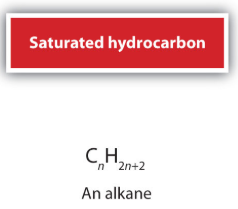
The general formula of alkenes is:
\( \mathrm{C_{n}H_{2n}} \) (for non-cyclic alkenes with one double bond)
- Compared to alkanes (\( \mathrm{C_{n}H_{2n+2}} \)), alkenes have fewer hydrogen atoms.
- This is due to the presence of a double bond.
Examples
- \( \mathrm{C_2H_4} \) → ethene
- \( \mathrm{C_3H_6} \) → propene
- \( \mathrm{C_4H_8} \) → butene
Cycloalkenes
Cycloalkenes are cyclic hydrocarbons that contain a carbon–carbon double bond.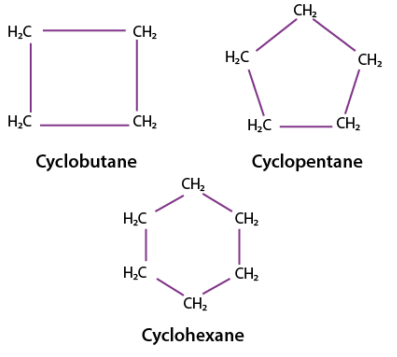
- Also have the general formula \( \mathrm{C_{n}H_{2n-2}} \) (due to ring + double bond).
- Example: cyclohexene → \( \mathrm{C_6H_{10}} \)
Unsaturation
Alkenes are described as unsaturated hydrocarbons.
- They contain at least one C=C double bond.
- Can undergo addition reactions (unlike alkanes).
Therefore, unsaturation refers to the presence of multiple bonds (not fully saturated with hydrogen).
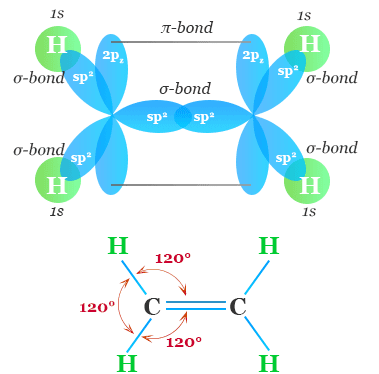
Structure of the Double Bond
A carbon–carbon double bond consists of two different types of bonds:
- σ (sigma) bond

- Formed by head-on overlap of orbitals.
- Strong bond along the axis between nuclei.
- π (pi) bond
- Formed by sideways overlap of p orbitals.
- Electron density above and below the bond axis.
- Weaker than the σ bond.
Therefore, a double bond = 1 σ bond + 1 π bond.
Key Consequences of the π Bond
- π bond is weaker → more reactive than alkanes.
- Electron-rich region → attracts electrophiles.
- Prevents free rotation around the C=C bond.
Summary
- Alkenes have general formula \( \mathrm{C_{n}H_{2n}} \).
- Cycloalkenes have formula \( \mathrm{C_{n}H_{2n-2}} \).
- They are unsaturated due to the presence of a C=C bond.
- Double bond consists of one σ bond and one π bond.
- π bond makes alkenes more reactive.
Therefore, the presence of the double bond determines both the structure and reactivity of alkenes.
Example 1 :
Explain why alkenes are more reactive than alkanes in terms of bonding.
▶️ Answer/Explanation
Alkenes contain a π bond in addition to a σ bond.
The π bond is weaker due to sideways overlap of p orbitals.
It has high electron density above and below the bond axis.
This makes it susceptible to attack by electrophiles.
Therefore, alkenes are more reactive than alkanes.
Example 2 :
Deduce the molecular formula of a cycloalkene with 7 carbon atoms and explain your reasoning.
▶️ Answer/Explanation
Cycloalkenes have the general formula \( \mathrm{C_{n}H_{2n-2}} \).
Substituting \( n = 7 \):
\( \mathrm{C_7H_{14-2} = C_7H_{12}} \)
The reduction in hydrogen atoms is due to one ring and one double bond.
Therefore, the molecular formula is \( \mathrm{C_7H_{12}} \).