Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.2 Geometric isomerism-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.2 Geometric isomerism- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.2 Geometric isomerism- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.2 be able to explain geometric isomerism in terms of restricted rotation around a C=C double bond and the nature of the substituents on the carbon atoms
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.2 Geometric (E/Z) Isomerism
Geometric isomerism is a type of stereoisomerism that arises due to restricted rotation around a carbon–carbon double bond and the arrangement of substituents attached to the double-bonded carbons.
Restricted Rotation Around C=C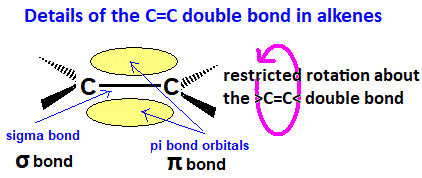
A C=C double bond consists of a σ bond and a π bond.
- The σ bond allows free rotation.
- The π bond prevents rotation because it would require breaking the bond.
Therefore, atoms attached to the double bond are fixed in position.
Condition for Geometric Isomerism
For geometric isomerism to exist:
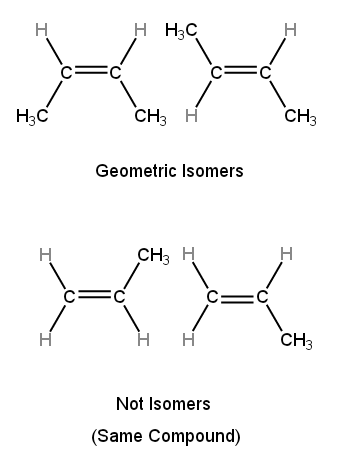
- Each carbon in the C=C bond must have two different substituents.
If this condition is not met, geometric isomerism is not possible.
Cis–Trans Isomerism (Basic Case)
When identical groups are present, isomers are described as cis and trans.
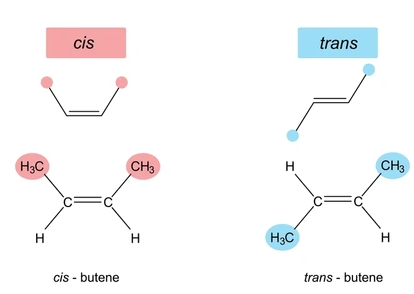
- cis → identical groups on the same side of the double bond
- trans → identical groups on opposite sides
Example: but-2-ene
- cis-but-2-ene → \( \mathrm{CH_3} \) groups on same side
- trans-but-2-ene → \( \mathrm{CH_3} \) groups on opposite sides
E/Z Isomerism (General Case)
When all substituents are different, E/Z notation is used instead of cis/trans.
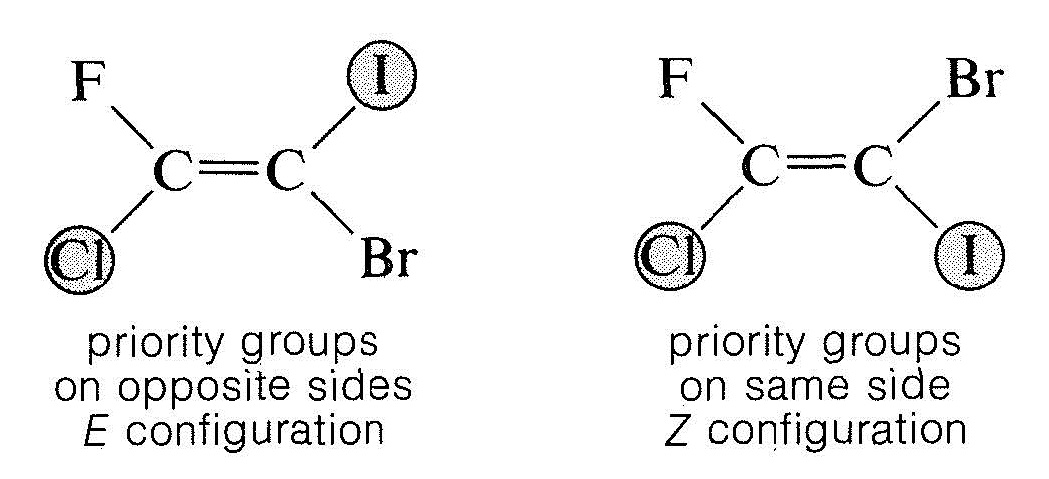
- Based on Cahn–Ingold–Prelog priority rules.
- Assign priority based on atomic number.
- Z (zusammen)
- Higher priority groups on the same side.
- E (entgegen)
- Higher priority groups on opposite sides.
Why Geometric Isomerism Occurs
- π bond prevents rotation.
- Different spatial arrangements become possible.
- These arrangements cannot interconvert without breaking the double bond.
Therefore, distinct isomers exist with different physical properties.
Effect on Properties
- Different boiling points.
- Different dipole moments.
- Different stability (trans usually more stable).
Summary
- Geometric isomerism arises due to restricted rotation around C=C.
- Requires two different groups on each carbon.
- Can be described using cis/trans or E/Z notation.
- Isomers have different spatial arrangements and properties.
Therefore, geometric isomerism is a direct consequence of the π bond restricting rotation and the nature of substituents.
Example 1:
Explain why \( \mathrm{CH_2=CHCl} \) does not show geometric isomerism.
▶️ Answer/Explanation
For geometric isomerism, each carbon must have two different substituents.
In \( \mathrm{CH_2=CHCl} \), one carbon has two identical hydrogen atoms.
Therefore, geometric isomerism is not possible.
Example 2:
Determine whether \( \mathrm{CH_3CH=CHBr} \) shows E/Z isomerism and justify your answer.
▶️ Answer/Explanation
Each carbon in the double bond has two different substituents.
One carbon: \( \mathrm{CH_3} \) and H.
Other carbon: Br and H.
Therefore, geometric isomerism is possible.
E/Z isomers can be assigned based on priority of substituents.
